Paper List
-
Autonomous Agents Coordinating Distributed Discovery Through Emergent Artifact Exchange
This paper addresses the fundamental limitation of current AI-assisted scientific research by enabling truly autonomous, decentralized investigation w...
-
D-MEM: Dopamine-Gated Agentic Memory via Reward Prediction Error Routing
This paper addresses the fundamental scalability bottleneck in LLM agentic memory systems: the O(N²) computational complexity and unbounded API token ...
-
Countershading coloration in blue shark skin emerges from hierarchically organized and spatially tuned photonic architectures inside skin denticles
This paper solves the core problem of how blue sharks achieve their striking dorsoventral countershading camouflage, revealing that coloration origina...
-
Human-like Object Grouping in Self-supervised Vision Transformers
This paper addresses the core challenge of quantifying how well self-supervised vision models capture human-like object grouping in natural scenes, br...
-
Hierarchical pp-Adic Framework for Gene Regulatory Networks: Theory and Stability Analysis
This paper addresses the core challenge of mathematically capturing the inherent hierarchical organization and multi-scale stability of gene regulator...
-
Towards unified brain-to-text decoding across speech production and perception
This paper addresses the core challenge of developing a unified brain-to-text decoding framework that works across both speech production and percepti...
-
Dual-Laws Model for a theory of artificial consciousness
This paper addresses the core challenge of developing a comprehensive, testable theory of consciousness that bridges biological and artificial systems...
-
Pulse desynchronization of neural populations by targeting the centroid of the limit cycle in phase space
This work addresses the core challenge of determining optimal pulse timing and intensity for desynchronizing pathological neural oscillations when the...
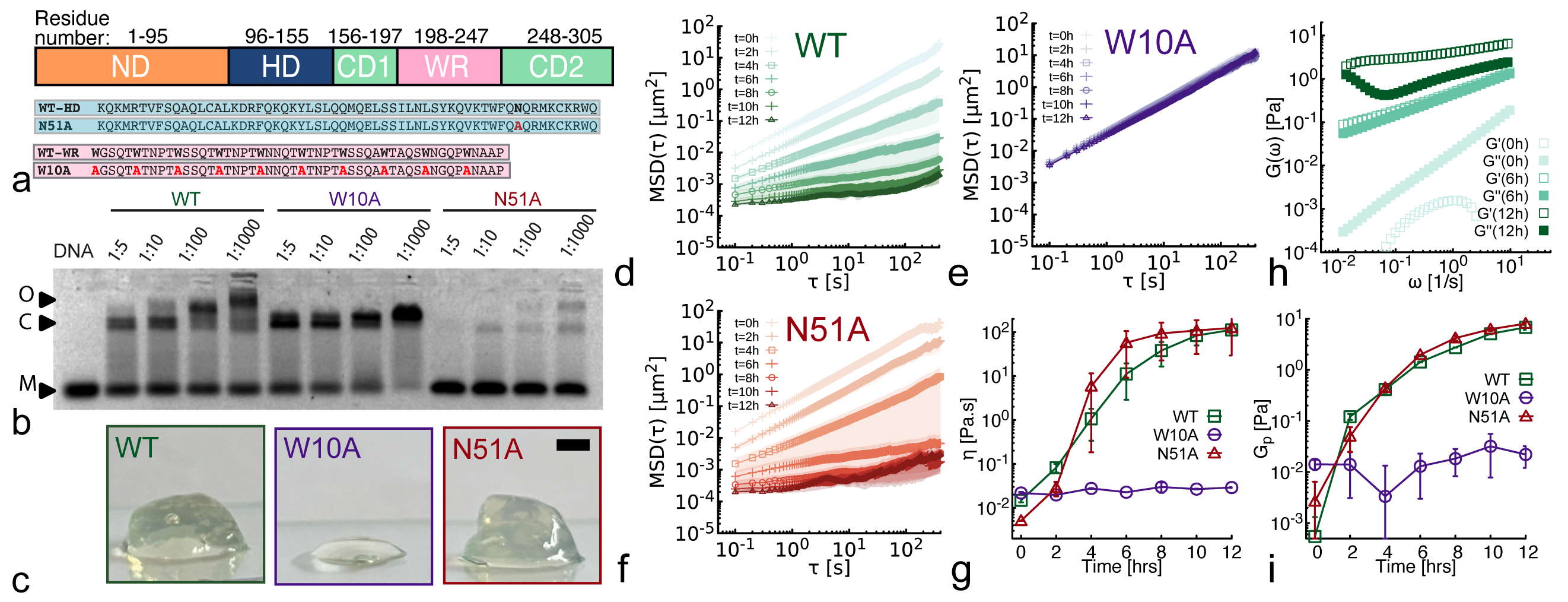
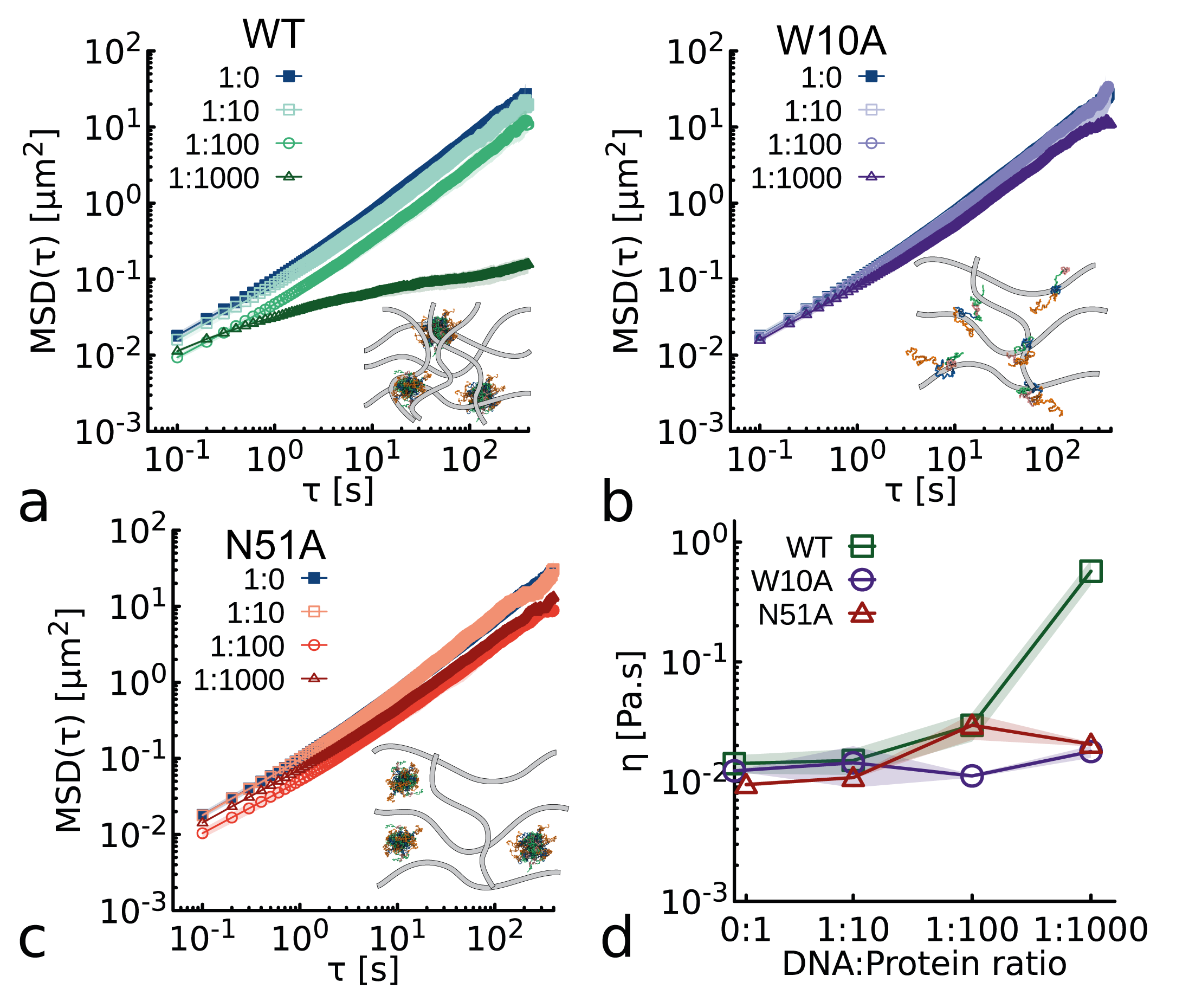
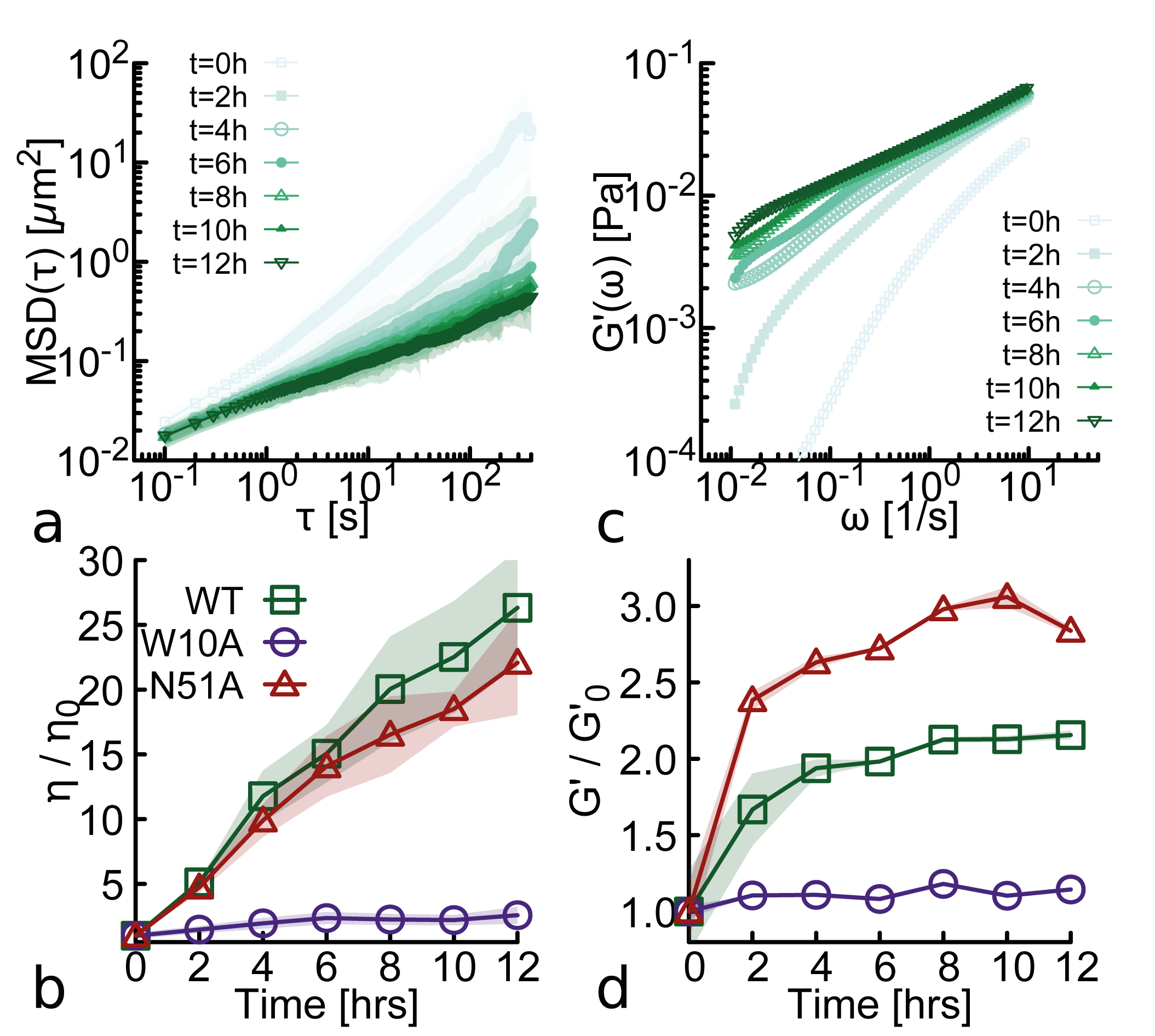
Modulation of DNA rheology by a transcription factor that forms aging microgels
University of Edinburgh | University of Glasgow | MRC Human Genetics Unit | WPI-SKCM2, Hiroshima University
30秒速读
IN SHORT: This work addresses the fundamental question of how the transcription factor NANOG, essential for embryonic stem cell pluripotency, physically regulates gene expression beyond simple DNA binding, by revealing its ability to form self-limiting, aging microgels that modulate DNA rheology.
核心创新
- Methodology First demonstration that a transcription factor (NANOG) forms self-limiting micelle-like clusters (~22-25 monomers) with exposed DNA-binding domains, acting as transient cross-linkers for DNA molecules.
- Biology Discovery of an aging microgel formation by NANOG, where viscoelasticity increases over time (10,000-fold viscosity increase over 12h), driven by its intrinsically disordered tryptophan-rich (WR) domain.
- Theory Proposes a novel 'rheological gene regulation' paradigm: NANOG may regulate gene expression not by large-scale chromatin reorganization, but by stabilizing and restricting the *dynamics* of key regulatory sites via aging condensates, potentially ingraining mechanical memory.
主要结论
- Wild-type NANOG forms macroscopic aging gels (10,000-fold viscosity increase over 12h at 37°C) and self-limiting micelle-like clusters (~22-25 proteins), while the oligomerization-deficient mutant (W10A) does not.
- Both clustering (via WR domain) and DNA binding (via homeodomain) are required for NANOG to act as an effective DNA cross-linker, significantly enhancing the viscoelasticity of entangled DNA solutions (observed in WT but not in W10A or DNA-binding-deficient N51A mutants).
- Aging (increasing viscoelasticity over time) occurs in NANOG-DNA solutions for both WT and the DNA-binding-deficient N51A mutant, indicating that oligomerization alone is sufficient to drive this slow restructuring toward gel-like states.
摘要: Proteins and nucleic acids form non-Newtonian liquids with complex rheological properties that contribute to their function in vivo. Here we investigate the rheology of the transcription factor NANOG, a key protein in sustaining embryonic stem cell self-renewal. We discover that at high concentrations NANOG forms macroscopic aging gels through its intrinsically disordered tryptophan-rich domain. By combining molecular dynamics simulations, mass photometry and Cryo-EM, we also discover that NANOG forms self-limiting micelle-like clusters which expose their DNA-binding domains. In dense solutions of DNA, NANOG micelle-like structures stabilize inter-molecular entanglements and crosslinks, forming microgel-like structures. Our findings suggest that NANOG may contribute to regulate gene expression in a unconventional way: by restricting and stabilizing genome dynamics at key transcriptional sites through the formation of an aging microgel-like structure, potentially enabling mechanical memory in the gene network.