Paper List
-
Evolutionarily Stable Stackelberg Equilibrium
通过要求追随者策略对突变入侵具有鲁棒性,弥合了斯塔克尔伯格领导力模型与演化稳定性之间的鸿沟。
-
Recovering Sparse Neural Connectivity from Partial Measurements: A Covariance-Based Approach with Granger-Causality Refinement
通过跨多个实验会话累积协方差统计,实现从部分记录到完整神经连接性的重建。
-
Atomic Trajectory Modeling with State Space Models for Biomolecular Dynamics
ATMOS通过提供一个基于SSM的高效框架,用于生物分子的原子级轨迹生成,弥合了计算昂贵的MD模拟与时间受限的深度生成模型之间的差距。
-
Slow evolution towards generalism in a model of variable dietary range
通过证明是种群统计噪声(而非确定性动力学)驱动了模式形成和泛化食性的演化,解决了间接竞争下物种形成的悖论。
-
Grounded Multimodal Retrieval-Augmented Drafting of Radiology Impressions Using Case-Based Similarity Search
通过将印象草稿基于检索到的历史病例,并采用明确引用和基于置信度的拒绝机制,解决放射学报告生成中的幻觉问题。
-
Unified Policy–Value Decomposition for Rapid Adaptation
通过双线性分解在策略和价值函数之间共享低维目标嵌入,实现对新颖任务的零样本适应。
-
Mathematical Modeling of Cancer–Bacterial Therapy: Analysis and Numerical Simulation via Physics-Informed Neural Networks
提供了一个严格的、无网格的PINN框架,用于模拟和分析细菌癌症疗法中复杂的、空间异质的相互作用。
-
Sample-Efficient Adaptation of Drug-Response Models to Patient Tumors under Strong Biological Domain Shift
通过从无标记分子谱中学习可迁移表征,利用最少的临床数据实现患者药物反应的有效预测。
The Effective Reproduction Number in the Kermack-McKendrick model with age of infection and reinfection
School of Mathematical Sciences, Beijing Normal University, Beijing 100875, People’s Republic of China.
30秒速读
IN SHORT: This paper addresses the challenge of accurately estimating the time-varying effective reproduction number ℛ(t) in epidemics by incorporating two critical real-world complexities: the age of infection (time since infection) and the possibility of reinfection.
核心创新
- Methodology Introduces a novel extension of the classical Kermack-McKendrick SIRS model by formally incorporating both infection-age structure (a) and a reinfection term (δ), moving beyond constant transmission rate assumptions.
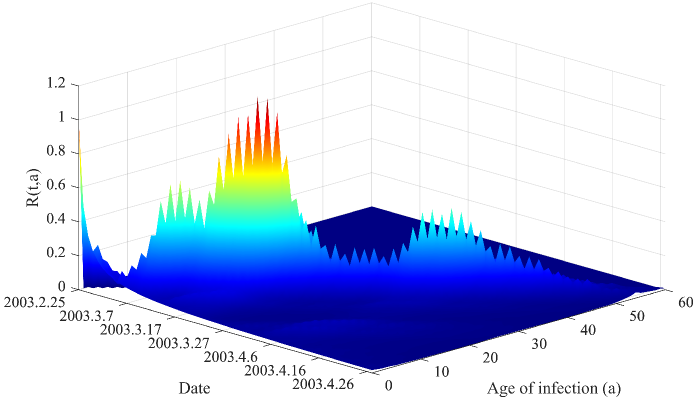
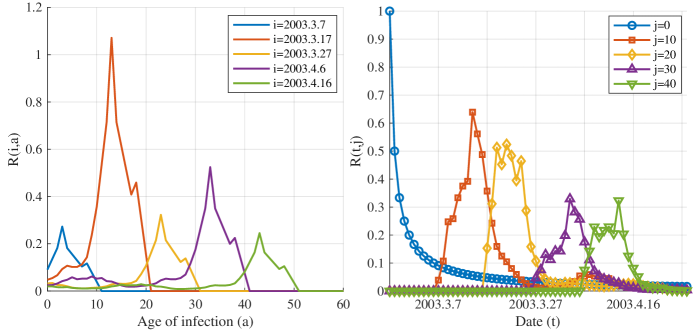
- Methodology Derives a rigorous mathematical framework using Volterra integral equations, the contraction mapping principle, and measure-valued solutions (e.g., Dirac mass for initial cohorts) to connect the flow of new infections N(t) to the reproductive power ℛ(t,a) and ultimately ℛ(t).
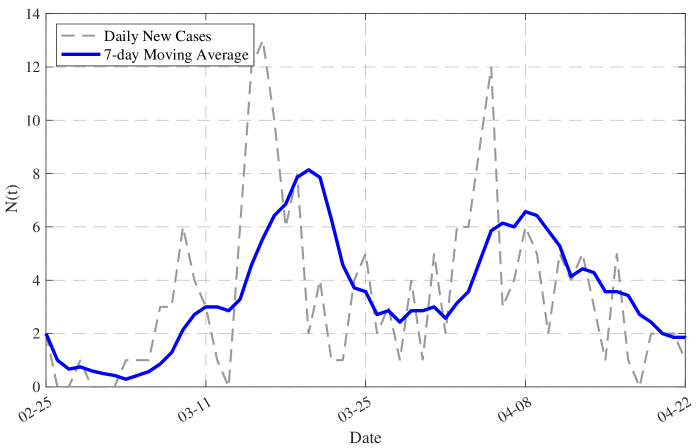
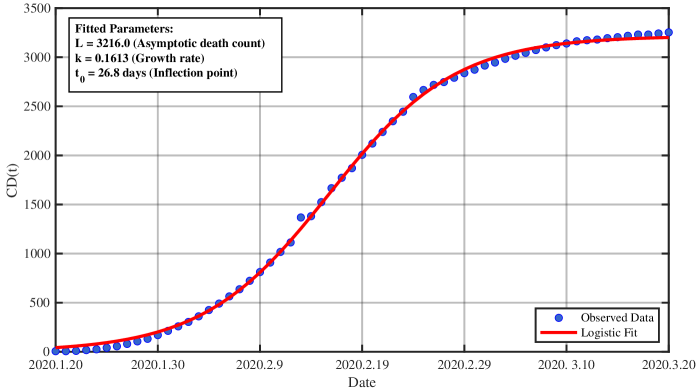
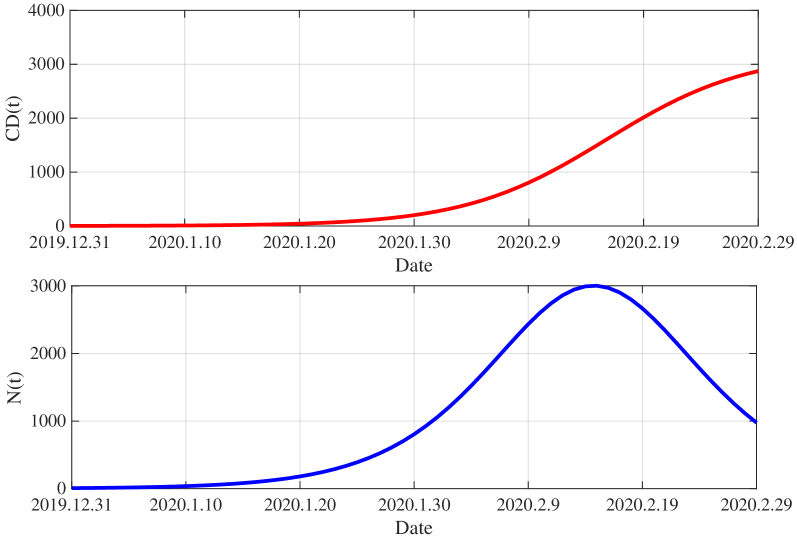
- Methodology/Biology Develops a practical parameter identification methodology that works with two common but challenging data types: 1) direct daily new case counts (applied to 2003 SARS in Singapore) and 2) cumulative death counts when new infection data is unreliable (applied to COVID-19 in China).
主要结论
- The model successfully formulates the infection dynamics as a nonlinear Volterra integral equation of the second kind for N(t) (Eq. 2.14), providing a solvable link between observable data and the underlying transmission parameters.
- Theoretical analysis justifies the use of a Dirac mass initial condition (representing a single cohort infected at time t0) via a limiting process of approximating functions i_κ(a), proving uniform convergence of the solution N_κ(t) to N(t) (Theorem 3.2).
- The derived framework enables the identification of the effective reproduction number ℛ(t) from epidemic curves, demonstrated through application to real-world SARS and COVID-19 datasets, bridging theoretical constructs with practical public health analytics.
摘要: This study introduces a novel epidemiological model that expands upon the Kermack-McKendrick model by incorporating the age of infection and reinfection. By including infection age, we can classify participants, which enables a more targeted analysis within the modeling framework. The reinfection term addresses the real-world occurrences of secondary or recurrent viral infections. In the theoretical part, we apply the contraction mapping principle, the dominated convergence theorem, and the properties of Volterra integral equations to derive analytical expressions for the number of newly infected individuals denoted by N(t). Then, we establish a Volterra integral equation for N(t) and study its initial conditions for both a single cohort and multiple cohorts. From this equation, we derive a method for identifying the effective reproduction number, denoted as ℛ(t). In the practical aspect, we present two distinct methods and separately apply them to analyze the daily new infection cases from the 2003 SARS outbreak in Singapore and the cumulative number of deaths from the COVID-19 epidemic in China. This work effectively bridges theoretical epidemiology and computational modeling, providing a robust framework for analyzing infection dynamics influenced by infection-age-structured transmission and reinfection mechanisms.