Paper List
-
Evolutionarily Stable Stackelberg Equilibrium
通过要求追随者策略对突变入侵具有鲁棒性,弥合了斯塔克尔伯格领导力模型与演化稳定性之间的鸿沟。
-
Recovering Sparse Neural Connectivity from Partial Measurements: A Covariance-Based Approach with Granger-Causality Refinement
通过跨多个实验会话累积协方差统计,实现从部分记录到完整神经连接性的重建。
-
Atomic Trajectory Modeling with State Space Models for Biomolecular Dynamics
ATMOS通过提供一个基于SSM的高效框架,用于生物分子的原子级轨迹生成,弥合了计算昂贵的MD模拟与时间受限的深度生成模型之间的差距。
-
Slow evolution towards generalism in a model of variable dietary range
通过证明是种群统计噪声(而非确定性动力学)驱动了模式形成和泛化食性的演化,解决了间接竞争下物种形成的悖论。
-
Grounded Multimodal Retrieval-Augmented Drafting of Radiology Impressions Using Case-Based Similarity Search
通过将印象草稿基于检索到的历史病例,并采用明确引用和基于置信度的拒绝机制,解决放射学报告生成中的幻觉问题。
-
Unified Policy–Value Decomposition for Rapid Adaptation
通过双线性分解在策略和价值函数之间共享低维目标嵌入,实现对新颖任务的零样本适应。
-
Mathematical Modeling of Cancer–Bacterial Therapy: Analysis and Numerical Simulation via Physics-Informed Neural Networks
提供了一个严格的、无网格的PINN框架,用于模拟和分析细菌癌症疗法中复杂的、空间异质的相互作用。
-
Sample-Efficient Adaptation of Drug-Response Models to Patient Tumors under Strong Biological Domain Shift
通过从无标记分子谱中学习可迁移表征,利用最少的临床数据实现患者药物反应的有效预测。
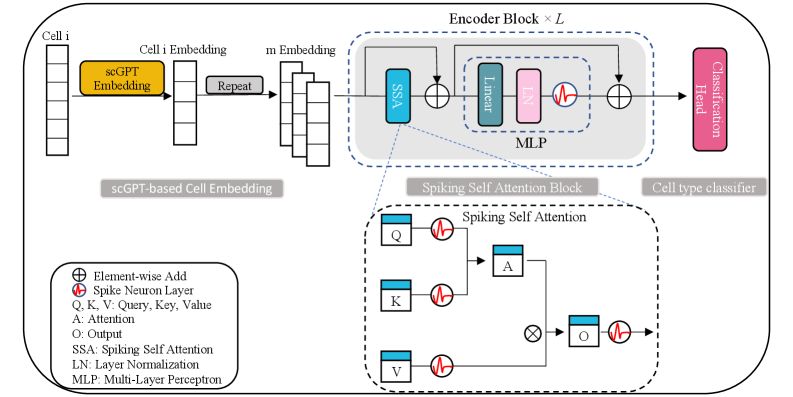
SpikGPT: A High-Accuracy and Interpretable Spiking Attention Framework for Single-Cell Annotation
Department of Biomedical Informatics, Emory University | Department of Surgery, Duke University
30秒速读
IN SHORT: This paper addresses the core challenge of robust single-cell annotation across heterogeneous datasets with batch effects and the critical need to identify previously unseen cell populations.
核心创新
- Methodology First integration of spiking neural networks with transformer architecture for single-cell analysis, using Leaky Integrate-and-Fire (LIF) neurons in a multi-head Spiking Self-Attention mechanism for energy-efficient computation.
- Methodology Novel two-step embedding expansion strategy: repeating cell embeddings along feature channels (default m=300) and temporal dimensions (default T=4) to enhance representation richness and training stability.
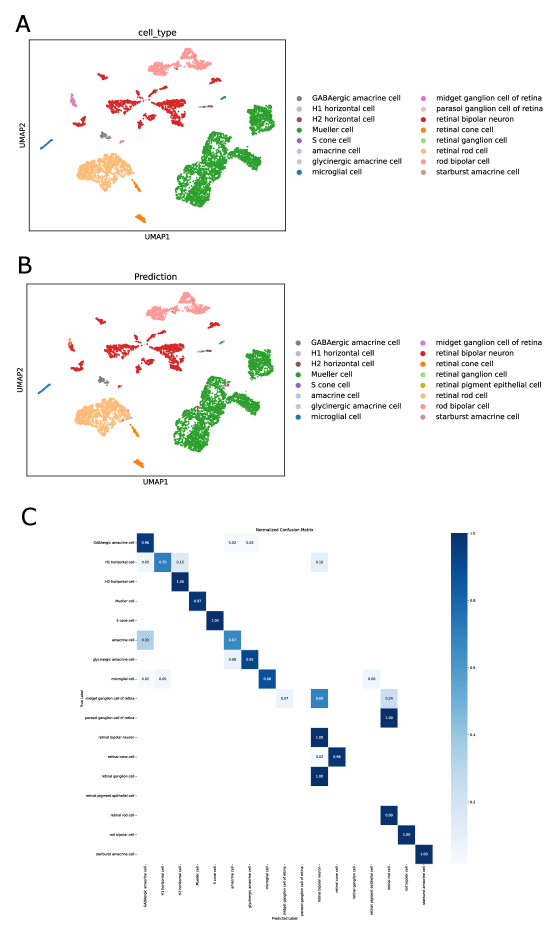
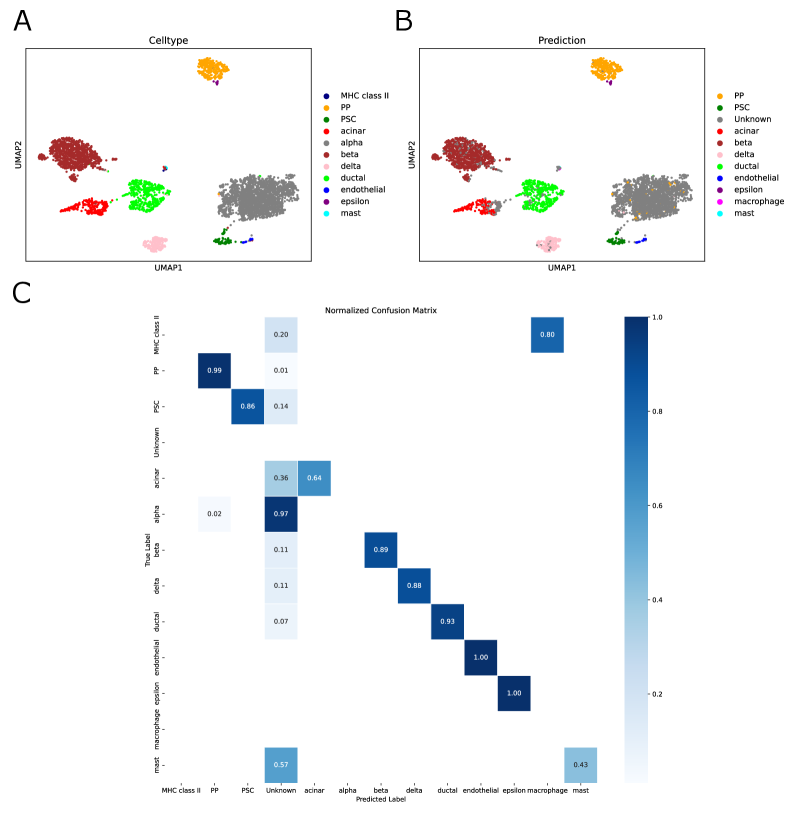
- Biology Confidence-based rejection mechanism that successfully identifies 97% of unseen 'alpha cells' as 'Unknown' in pancreas datasets, enabling robust detection of novel cell types absent from training data.
主要结论
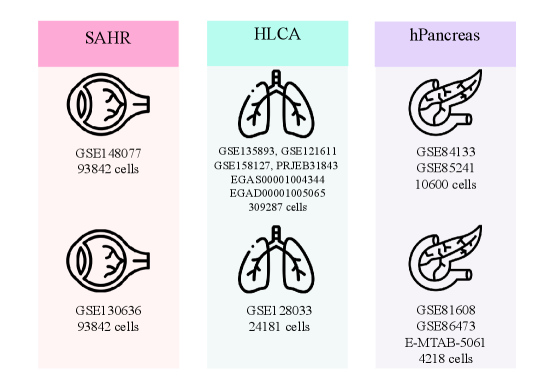
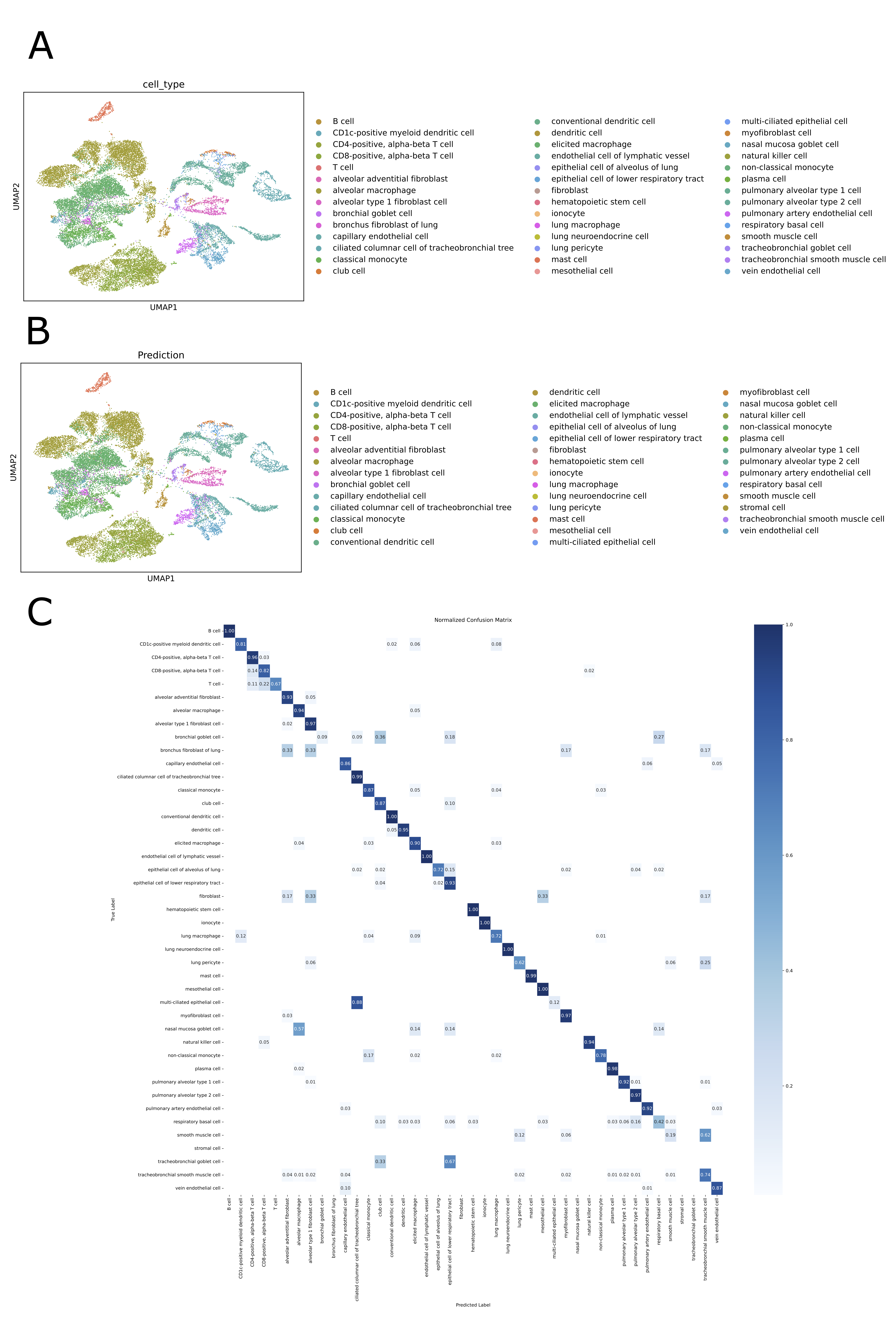
- SpikGPT achieves accuracy of 0.991 on SAHR dataset and 0.920 on HLCA dataset, outperforming or matching 8 benchmark methods including scGPT, CCA, and scPred.
- The model demonstrates superior robustness to batch effects, maintaining macro F1-score of 0.711 on heterogeneous HLCA data where traditional methods like SingleR drop to 0.207 F1-score.
- SpikGPT successfully identifies 97% of unseen 'alpha cells' as 'Unknown' using confidence thresholding (p<0.05), enabling reliable detection of novel cell populations.
摘要: Accurate and scalable cell type annotation remains a challenge in single-cell transcriptomics, especially when datasets exhibit strong batch effects or contain previously unseen cell populations. Here we introduce SpikGPT, a hybrid deep learning framework that integrates scGPT-derived cell embeddings with a spiking Transformer architecture to achieve efficient and robust annotation. scGPT provides biologically informed dense representations of each cell, which are further processed by a multi-head Spiking Self-Attention mechanism, energy-efficient feature extraction. Across multiple benchmark datasets, SpikGPT consistently matches or exceeds the performance of leading annotation tools. Notably, SpikGPT uniquely identifies unseen cell types by assigning low-confidence predictions to an 'Unknown' category, allowing accurate rejection of cell states absent from the training reference. Together, these results demonstrate that SpikGPT is a versatile and reliable annotation tool capable of generalizing across datasets, resolving complex cellular heterogeneity, and facilitating discovery of novel or disease-associated cell populations.