Paper List
-
Discovery of a Hematopoietic Manifold in scGPT Yields a Method for Extracting Performant Algorithms from Biological Foundation Model Internals
This work addresses the core challenge of extracting reusable, interpretable, and high-performance biological algorithms from the opaque internal repr...
-
MS2MetGAN: Latent-space adversarial training for metabolite–spectrum matching in MS/MS database search
This paper addresses the critical bottleneck in metabolite identification: the generation of high-quality negative training samples that are structura...
-
Toward Robust, Reproducible, and Widely Accessible Intracranial Language Brain-Computer Interfaces: A Comprehensive Review of Neural Mechanisms, Hardware, Algorithms, Evaluation, Clinical Pathways and Future Directions
This review addresses the core challenge of fragmented and heterogeneous evidence that hinders the clinical translation of intracranial language BCIs,...
-
Less Is More in Chemotherapy of Breast Cancer
通过纳入细胞周期时滞和竞争项,解决了现有肿瘤-免疫模型的过度简化问题,以定量比较化疗方案。
-
Fold-CP: A Context Parallelism Framework for Biomolecular Modeling
This paper addresses the critical bottleneck of GPU memory limitations that restrict AlphaFold 3-like models to processing only a few thousand residue...
-
Open Biomedical Knowledge Graphs at Scale: Construction, Federation, and AI Agent Access with Samyama Graph Database
This paper addresses the core pain point of fragmented biomedical data by constructing and federating large-scale, open knowledge graphs to enable sea...
-
Predictive Analytics for Foot Ulcers Using Time-Series Temperature and Pressure Data
This paper addresses the critical need for continuous, real-time monitoring of diabetic foot health by developing an unsupervised anomaly detection fr...
-
Hypothesis-Based Particle Detection for Accurate Nanoparticle Counting and Digital Diagnostics
This paper addresses the core challenge of achieving accurate, interpretable, and training-free nanoparticle counting in digital diagnostic assays, wh...
Mapping of Lesion Images to Somatic Mutations
University of Illinois at Chicago | University of Texas MD Anderson Cancer Center
30秒速读
IN SHORT: This paper addresses the critical bottleneck of delayed genetic analysis in cancer diagnosis by predicting a patient's full somatic mutation profile directly from medical lesion images, enabling earlier targeted treatment decisions.
核心创新
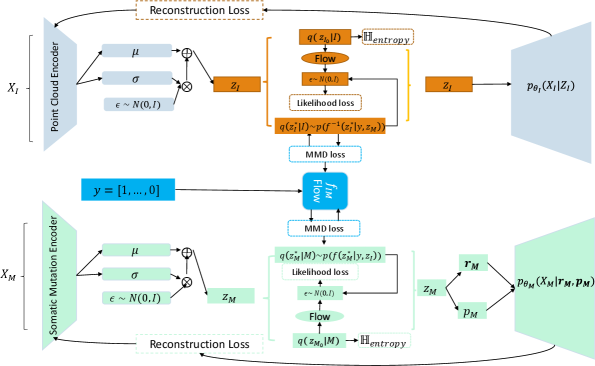
- Methodology Proposes LLOST, a novel architecture with dual VAEs and a separate, cancer-type-conditioned shared latent space, coupled with domain-specific conditional Normalizing Flow priors to handle heterogeneous data distributions.
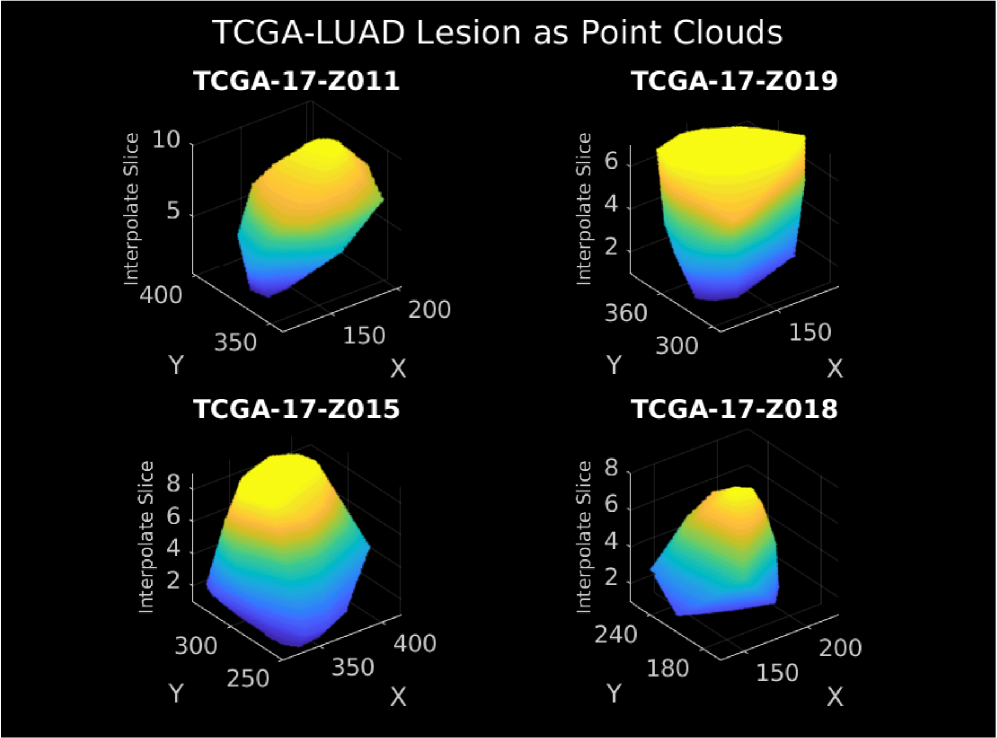
- Methodology Introduces a modality-invariant point cloud representation for lesion images, overcoming challenges of multi-slice, multi-modal (CT/MRI) medical imaging data.
- Methodology Employs a Negative-Binomial likelihood within the mutation VAE to effectively model the high-dimensional, sparse, and discrete nature of somatic mutation count data.
主要结论
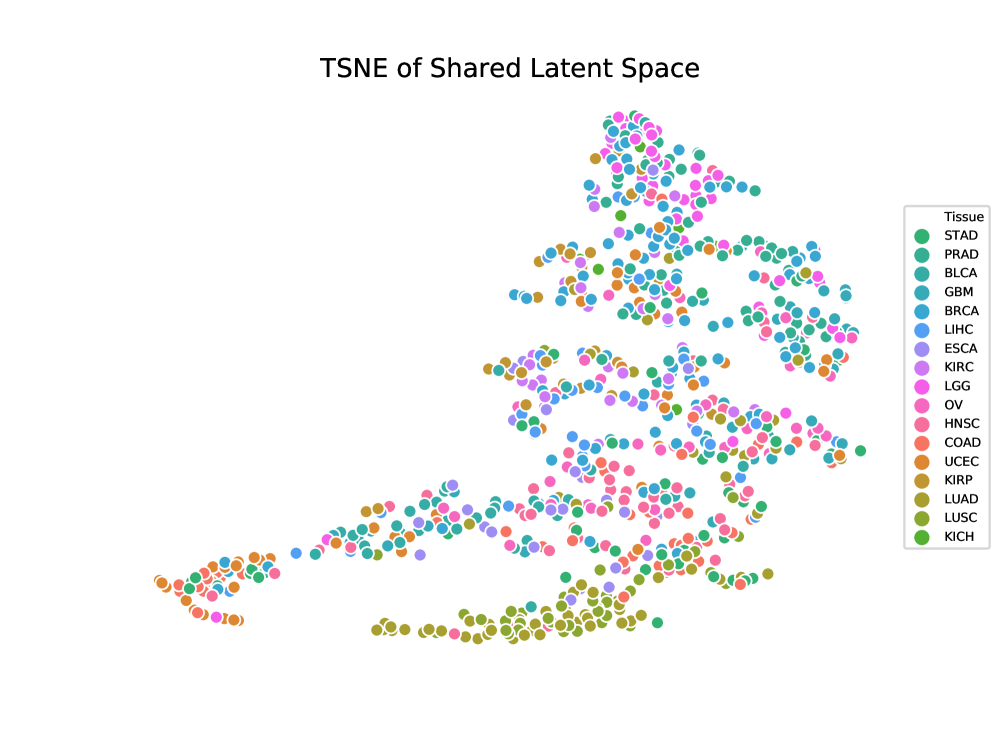
- LLOST successfully learns a shared latent representation between lesion point clouds and somatic mutation counts, capturing cancer-type-specific patterns across these disparate domains.
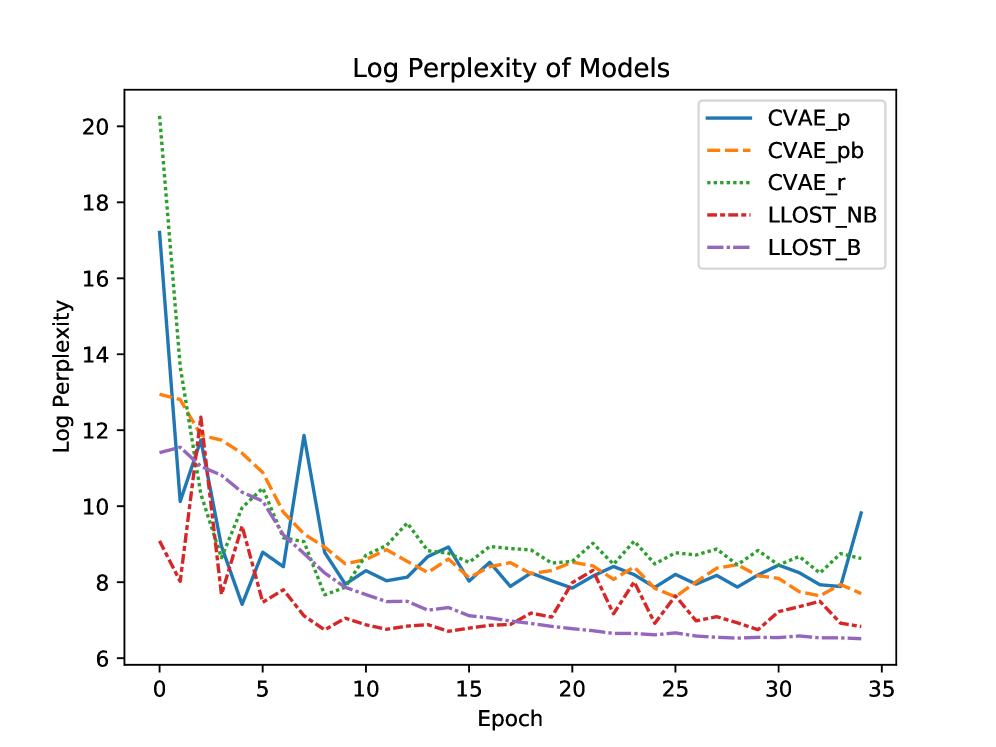
- The model demonstrates predictive capability for both mutation occurrence (binary prediction) and mutation counts, validated on a dataset of 1342 patients across 18 cancer types from TCGA/TCIA.
- The use of conditional Normalizing Flow priors and a separate shared latent space allows the model to account for and bridge the complex, distinct distributions of imaging and genomic data.
摘要: Medical imaging is a critical initial tool used by clinicians to determine a patient’s cancer diagnosis, allowing for faster intervention and more reliable patient prognosis. At subsequent stages of patient diagnosis, genetic information is extracted to help select specific patient treatment options. As the efficacy of cancer treatment often relies on early diagnosis and treatment, we build a deep latent variable model to determine patients’ somatic mutation profiles based on their corresponding medical images. We first introduce a point cloud representation of lesions images to allow for invariance to the imaging modality. We then propose, LLOST, a model with dual variational autoencoders coupled together by a separate shared latent space that unifies features from the lesion point clouds and counts of distinct somatic mutations. Therefore our model consists of three latent space, each of which is learned with a conditional normalizing flow prior to account for the diverse distributions of each domain. We conduct qualitative and quantitative experiments on de-identified medical images from The Cancer Imaging Archive and the corresponding somatic mutations from the Pan Cancer dataset of The Cancer Genomic Archive. We show the model’s predictive performance on the counts of specific mutations as well as it’s ability to accurately predict the occurrence of mutations. In particular, shared patterns between the imaging and somatic mutation domain that reflect cancer type. We conclude with a remark on how to improve the model and possible future avenues of research to include other genetic domains.