Paper List
-
Nyxus: A Next Generation Image Feature Extraction Library for the Big Data and AI Era
This paper addresses the core pain point of efficiently extracting standardized, comparable features from massive (terabyte to petabyte-scale) biomedi...
-
Topological Enhancement of Protein Kinetic Stability
This work addresses the long-standing puzzle of why knotted proteins exist by demonstrating that deep knots provide a functional advantage through enh...
-
A Multi-Label Temporal Convolutional Framework for Transcription Factor Binding Characterization
This paper addresses the critical limitation of existing TF binding prediction methods that treat transcription factors as independent entities, faili...
-
Social Distancing Equilibria in Games under Conventional SI Dynamics
This paper solves the core problem of proving the existence and uniqueness of Nash equilibria in finite-duration SI epidemic games, showing they are a...
-
Binding Free Energies without Alchemy
This paper addresses the core bottleneck of computational expense in Absolute Binding Free Energy calculations by eliminating the need for numerous al...
-
SHREC: A Spectral Embedding-Based Approach for Ab-Initio Reconstruction of Helical Molecules
This paper addresses the core bottleneck in cryo-EM helical reconstruction: eliminating the dependency on accurate initial symmetry parameter estimati...
-
Budget-Sensitive Discovery Scoring: A Formally Verified Framework for Evaluating AI-Guided Scientific Selection
This paper addresses the critical gap in evaluating AI-guided scientific selection strategies under realistic budget constraints, where existing metri...
-
Probabilistic Joint and Individual Variation Explained (ProJIVE) for Data Integration
This paper addresses the core challenge of accurately decomposing shared (joint) and dataset-specific (individual) sources of variation in multi-modal...
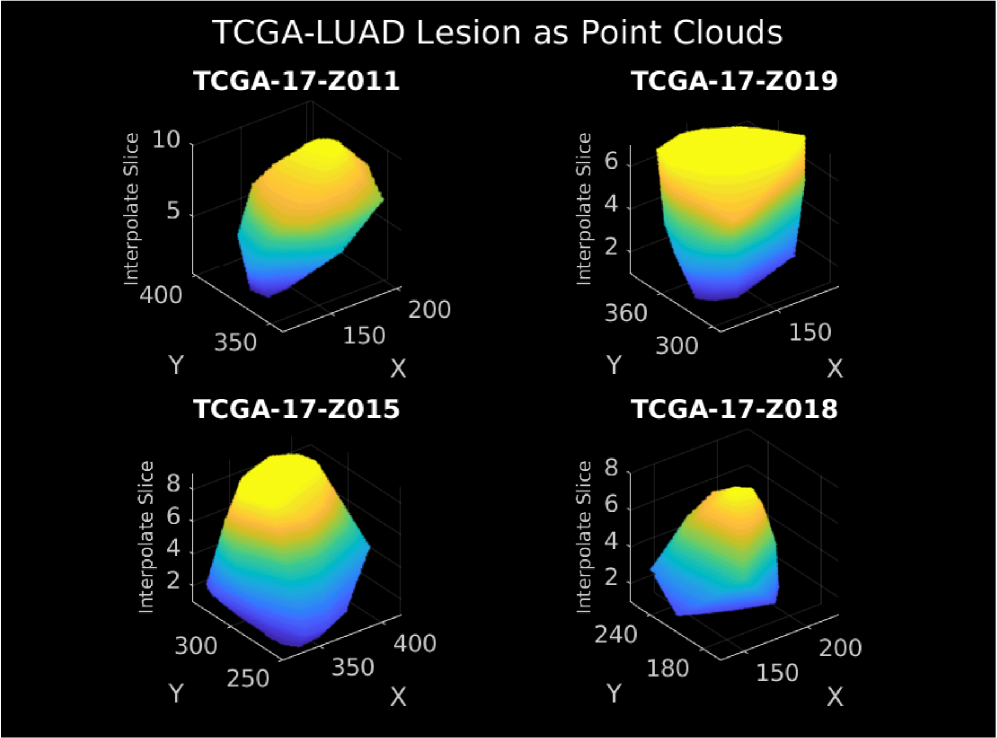
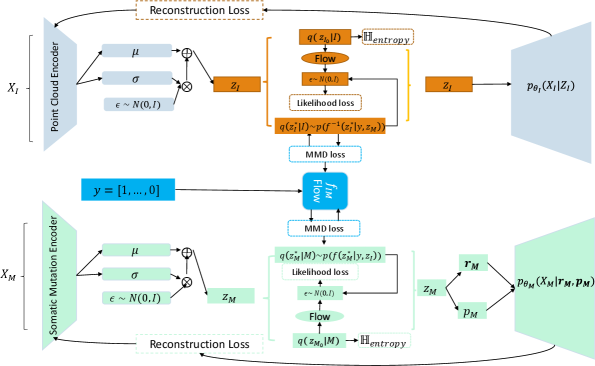
Mapping of Lesion Images to Somatic Mutations
University of Illinois at Chicago | University of Texas MD Anderson Cancer Center
30秒速读
IN SHORT: This paper addresses the critical bottleneck of delayed genetic analysis in cancer diagnosis by predicting a patient's full somatic mutation profile directly from medical lesion images, enabling earlier targeted treatment decisions.
核心创新
- Methodology Proposes LLOST, a novel architecture with dual VAEs and a separate, cancer-type-conditioned shared latent space, coupled with domain-specific conditional Normalizing Flow priors to handle heterogeneous data distributions.
- Methodology Introduces a modality-invariant point cloud representation for lesion images, overcoming challenges of multi-slice, multi-modal (CT/MRI) medical imaging data.
- Methodology Employs a Negative-Binomial likelihood within the mutation VAE to effectively model the high-dimensional, sparse, and discrete nature of somatic mutation count data.
主要结论
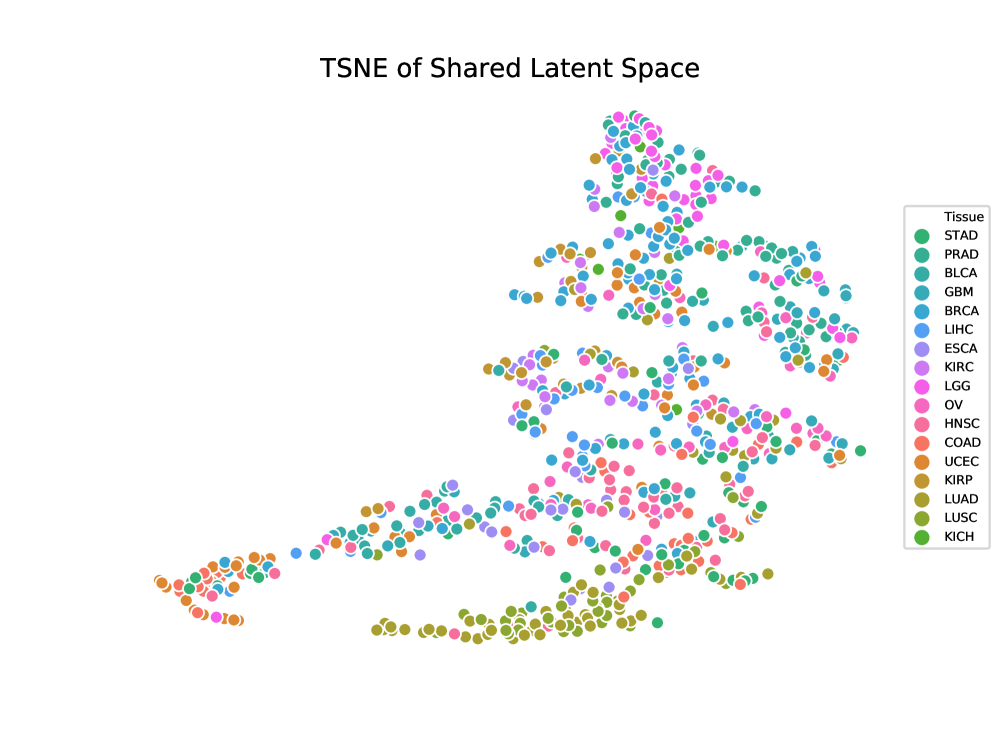
- LLOST successfully learns a shared latent representation between lesion point clouds and somatic mutation counts, capturing cancer-type-specific patterns across these disparate domains.
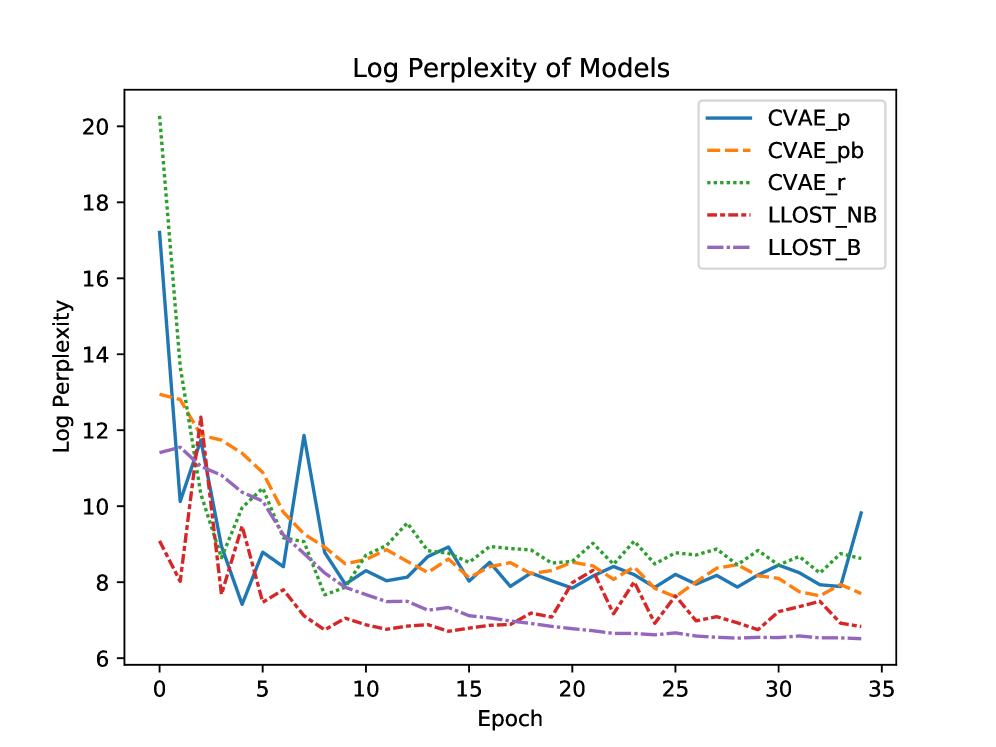
- The model demonstrates predictive capability for both mutation occurrence (binary prediction) and mutation counts, validated on a dataset of 1342 patients across 18 cancer types from TCGA/TCIA.
- The use of conditional Normalizing Flow priors and a separate shared latent space allows the model to account for and bridge the complex, distinct distributions of imaging and genomic data.
摘要: Medical imaging is a critical initial tool used by clinicians to determine a patient’s cancer diagnosis, allowing for faster intervention and more reliable patient prognosis. At subsequent stages of patient diagnosis, genetic information is extracted to help select specific patient treatment options. As the efficacy of cancer treatment often relies on early diagnosis and treatment, we build a deep latent variable model to determine patients’ somatic mutation profiles based on their corresponding medical images. We first introduce a point cloud representation of lesions images to allow for invariance to the imaging modality. We then propose, LLOST, a model with dual variational autoencoders coupled together by a separate shared latent space that unifies features from the lesion point clouds and counts of distinct somatic mutations. Therefore our model consists of three latent space, each of which is learned with a conditional normalizing flow prior to account for the diverse distributions of each domain. We conduct qualitative and quantitative experiments on de-identified medical images from The Cancer Imaging Archive and the corresponding somatic mutations from the Pan Cancer dataset of The Cancer Genomic Archive. We show the model’s predictive performance on the counts of specific mutations as well as it’s ability to accurately predict the occurrence of mutations. In particular, shared patterns between the imaging and somatic mutation domain that reflect cancer type. We conclude with a remark on how to improve the model and possible future avenues of research to include other genetic domains.