Paper List
-
Formation of Artificial Neural Assemblies by Biologically Plausible Inhibition Mechanisms
This work addresses the core limitation of the Assembly Calculus model—its fixed-size, biologically implausible k-WTA selection process—by introducing...
-
How to make the most of your masked language model for protein engineering
This paper addresses the critical bottleneck of efficiently sampling high-quality, diverse protein sequences from Masked Language Models (MLMs) for pr...
-
Module control in youth symptom networks across COVID-19
This paper addresses the core challenge of distinguishing whether a prolonged societal stressor (COVID-19) fundamentally reorganizes the architecture ...
-
JEDI: Jointly Embedded Inference of Neural Dynamics
This paper addresses the core challenge of inferring context-dependent neural dynamics from noisy, high-dimensional recordings using a single unified ...
-
ATP Level and Phosphorylation Free Energy Regulate Trigger-Wave Speed and Critical Nucleus Size in Cellular Biochemical Systems
This work addresses the core challenge of quantitatively predicting how the cellular energy state (ATP level and phosphorylation free energy) governs ...
-
Packaging Jupyter notebooks as installable desktop apps using LabConstrictor
This paper addresses the core pain point of ensuring Jupyter notebook reproducibility and accessibility across different computing environments, parti...
-
SNPgen: Phenotype-Supervised Genotype Representation and Synthetic Data Generation via Latent Diffusion
This paper addresses the core challenge of generating privacy-preserving synthetic genotype data that maintains both statistical fidelity and downstre...
-
Continuous Diffusion Transformers for Designing Synthetic Regulatory Elements
This paper addresses the challenge of efficiently generating novel, cell-type-specific regulatory DNA sequences with high predicted activity while min...
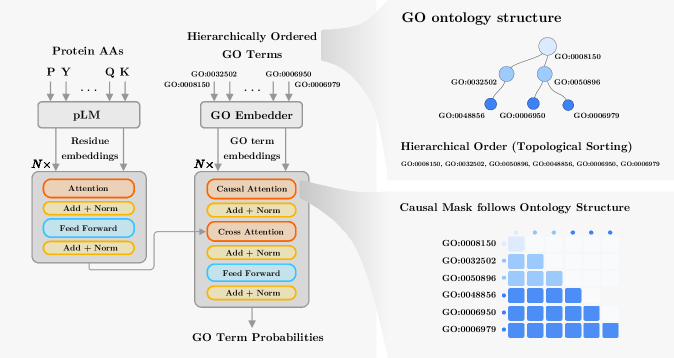
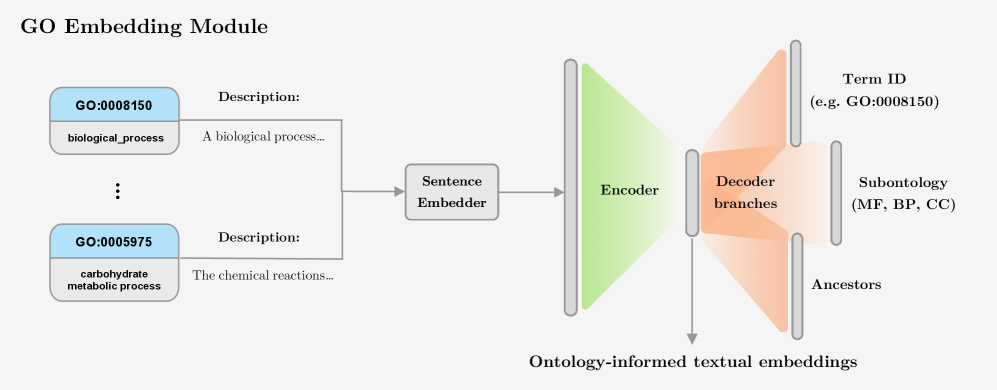
STAR-GO: Improving Protein Function Prediction by Learning to Hierarchically Integrate Ontology-Informed Semantic Embeddings
Department of Computer Engineering, Bogazici University, Istanbul, Turkiye
30秒速读
IN SHORT: This paper addresses the core challenge of generalizing protein function prediction to unseen or newly introduced Gene Ontology (GO) terms by overcoming the limitations of existing models that either prioritize graph structure at the expense of semantic meaning or vice versa.
核心创新
- Methodology Introduces a novel GO embedding module that integrates textual definitions (via SBERT-BioBERT) with ontology graph structure through a multi-task autoencoder, learning unified representations that preserve both semantic similarity and hierarchical dependencies.
- Methodology Proposes a hierarchical Transformer decoder that processes GO terms in topological order (ancestors to descendants) using causal self-attention, enabling information propagation across ontology levels and capturing functional dependencies.
- Biology Demonstrates superior zero-shot generalization to unseen GO terms, particularly for Molecular Function and Biological Process terms, by effectively leveraging semantic information from textual definitions, which transfers better to novel ontology concepts than purely structural embeddings.
主要结论
- STAR-GO achieves state-of-the-art or competitive performance across all three GO subontologies (BP, CC, MF), with the highest AUC scores (e.g., 0.989 for BP, 0.988 for CC, 0.995 for MF), indicating strong term-level discriminability.
- In zero-shot evaluation on 16 held-out GO terms, STAR-GO variants achieve the highest AUCs in 13 cases, significantly outperforming baselines like DeepGOZero and DeepGO-SE, demonstrating superior generalization to unseen functions.
- Ablation studies reveal that semantic embeddings (STAR_T) achieve the best zero-shot results for most MF and BP terms (e.g., AUC of 0.949 for GO:0001228), while structural embeddings (STAR_S) perform best for a few terms but poorly for MF, highlighting the critical role of semantic information for generalization.
摘要: Motivation: Accurate prediction of protein function is essential for elucidating molecular mechanisms and advancing biological and therapeutic discovery. Yet experimental annotation lags far behind the rapid growth of protein sequence data. Computational approaches address this gap by associating proteins with Gene Ontology (GO) terms, which encode functional knowledge through hierarchical relations and textual definitions. However, existing models often emphasize one modality over the other, limiting their ability to generalize, particularly to unseen or newly introduced GO terms that frequently arise as the ontology evolves, and making the previously trained models outdated. Results: We present STAR-GO, a Transformer-based framework that jointly models the semantic and structural characteristics of GO terms to enhance zero-shot protein function prediction. STAR-GO integrates textual definitions with ontology graph structure to learn unified GO representations, which are processed in hierarchical order to propagate information from general to specific terms. These representations are then aligned with protein sequence embeddings to capture sequence–function relationships. STAR-GO achieves state-of-the-art performance and superior zero-shot generalization, demonstrating the utility of integrating semantics and structure for robust and adaptable protein function prediction. Availability: Code and pre-trained models are available at https://github.com/boun-tabi-lifelu/stargo.