Paper List
-
The Effective Reproduction Number in the Kermack-McKendrick model with age of infection and reinfection
This paper addresses the challenge of accurately estimating the time-varying effective reproduction number ℛ(t) in epidemics by incorporating two crit...
-
Covering Relations in the Poset of Combinatorial Neural Codes
This work addresses the core challenge of navigating the complex poset structure of neural codes to systematically test the conjecture linking convex ...
-
Collective adsorption of pheromones at the water-air interface
This paper addresses the core challenge of understanding how amphiphilic pheromones, previously assumed to be transported in the gas phase, can be sta...
-
pHapCompass: Probabilistic Assembly and Uncertainty Quantification of Polyploid Haplotype Phase
This paper addresses the core challenge of accurately assembling polyploid haplotypes from sequencing data, where read assignment ambiguity and an exp...
-
Setting up for failure: automatic discovery of the neural mechanisms of cognitive errors
This paper addresses the core challenge of automating the discovery of biologically plausible recurrent neural network (RNN) dynamics that can replica...
-
Influence of Object Affordance on Action Language Understanding: Evidence from Dynamic Causal Modeling Analysis
This study addresses the core challenge of moving beyond correlational evidence to establish the *causal direction* and *temporal dynamics* of how obj...
-
Revealing stimulus-dependent dynamics through statistical complexity
This paper addresses the core challenge of detecting stimulus-specific patterns in neural population dynamics that remain hidden to traditional variab...
-
Exactly Solvable Population Model with Square-Root Growth Noise and Cell-Size Regulation
This paper addresses the fundamental gap in understanding how microscopic growth fluctuations, specifically those with size-dependent (square-root) no...
EnzyCLIP: A Cross-Attention Dual Encoder Framework with Contrastive Learning for Predicting Enzyme Kinetic Constants
Vellore Institute of Technology | BIT (Department of Computer Science) | BIT (Department of Bioengineering and Biotechnology)
30秒速读
IN SHORT: This paper addresses the core challenge of jointly predicting enzyme kinetic parameters (Kcat and Km) by modeling dynamic enzyme-substrate interactions through a multimodal contrastive learning framework.
核心创新
- Methodology Proposes a CLIP-inspired dual-encoder architecture with bidirectional cross-attention that dynamically models enzyme-substrate interactions, overcoming the limitation of separate processing in existing methods.
- Methodology Integrates contrastive learning (InfoNCE loss) with multi-task regression (Huber loss) to learn aligned multimodal representations while jointly predicting both Kcat and Km parameters.
- Biology Addresses the critical gap in existing literature that typically focuses on single parameter prediction (mainly Kcat) by providing a unified framework for joint prediction of both fundamental kinetic constants.
主要结论
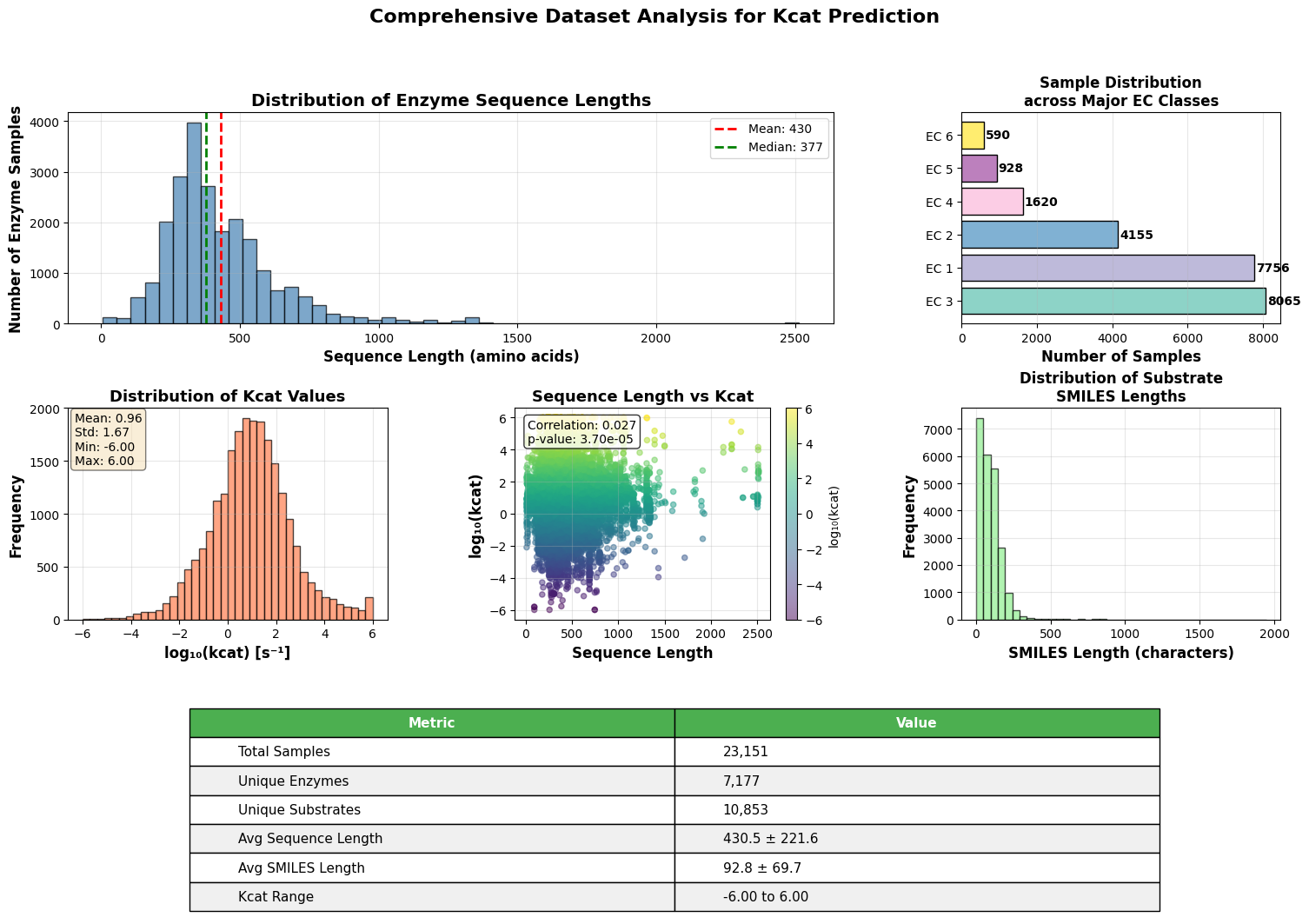
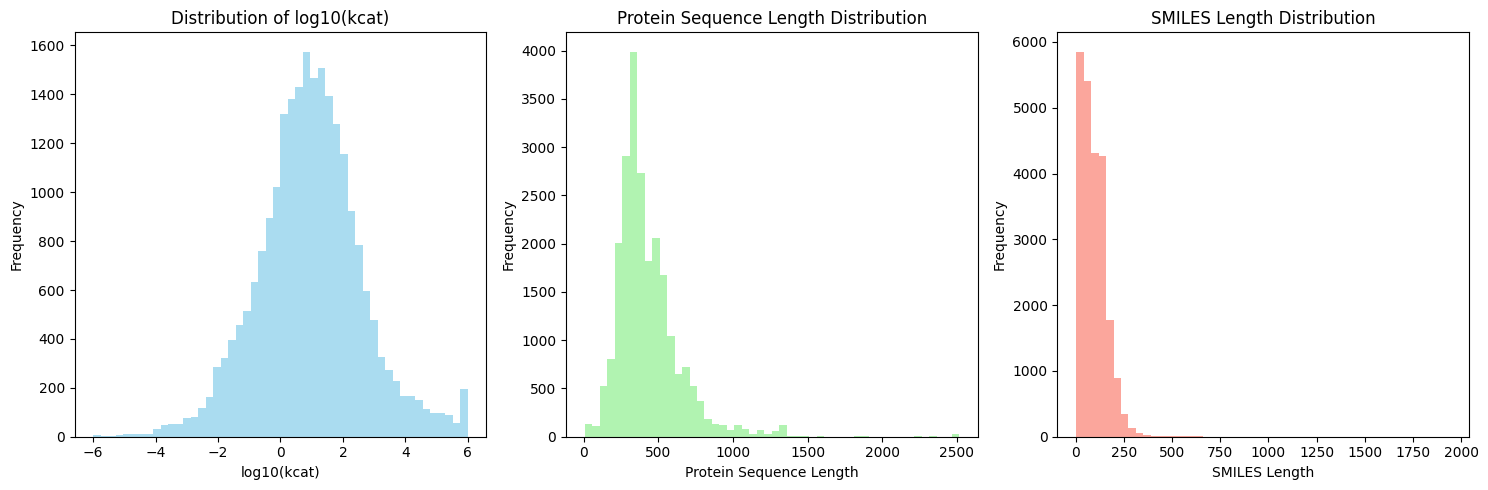
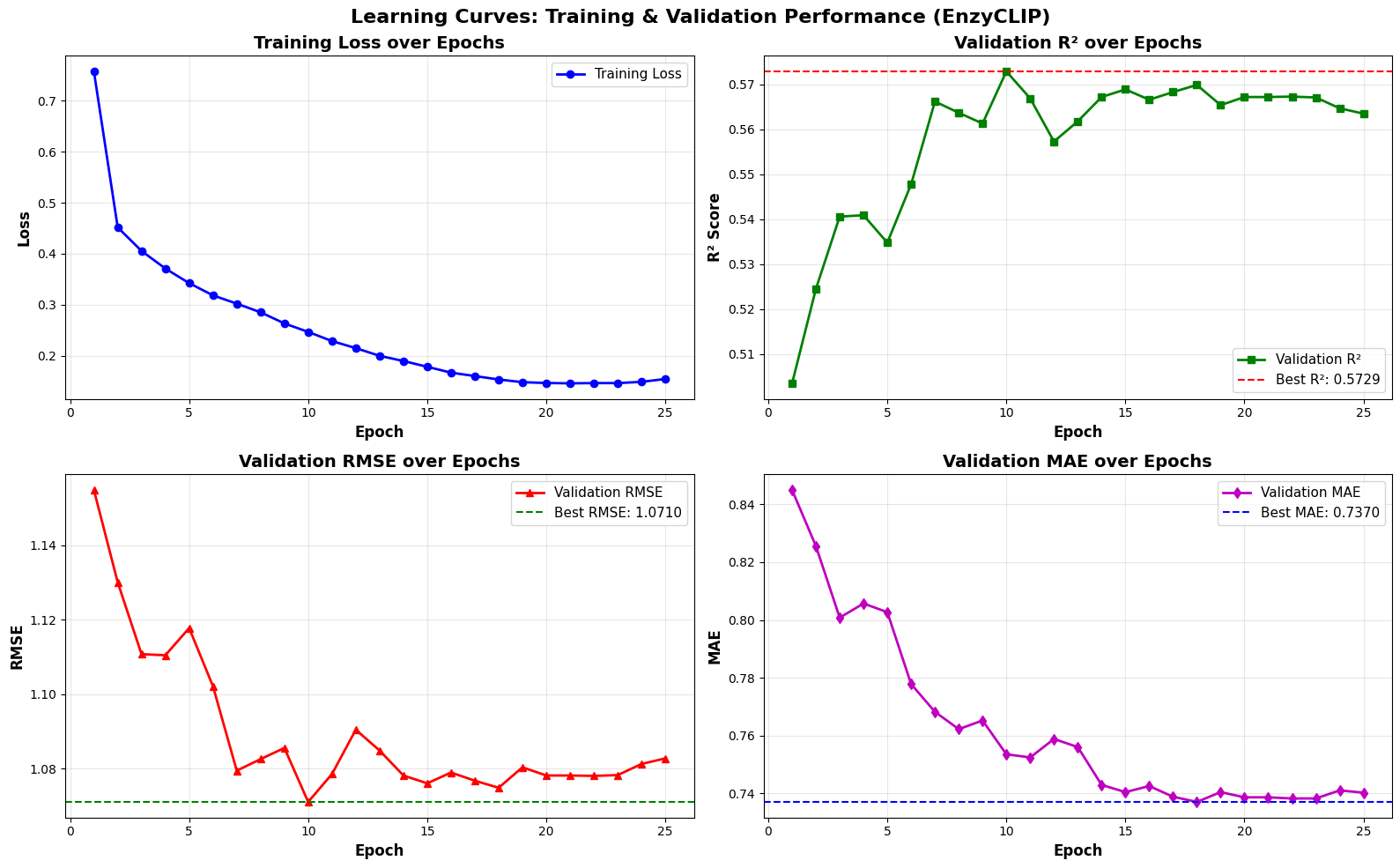
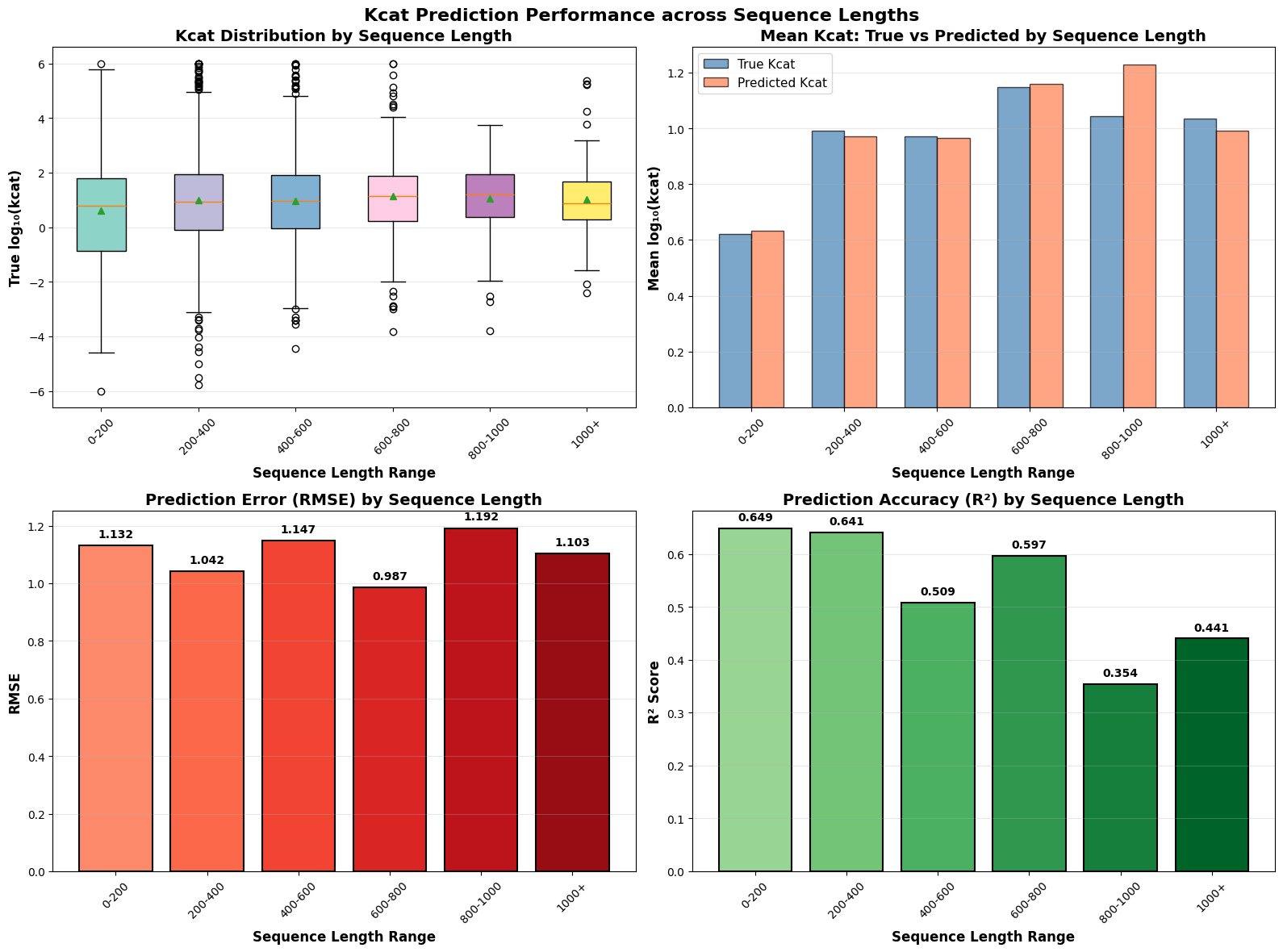
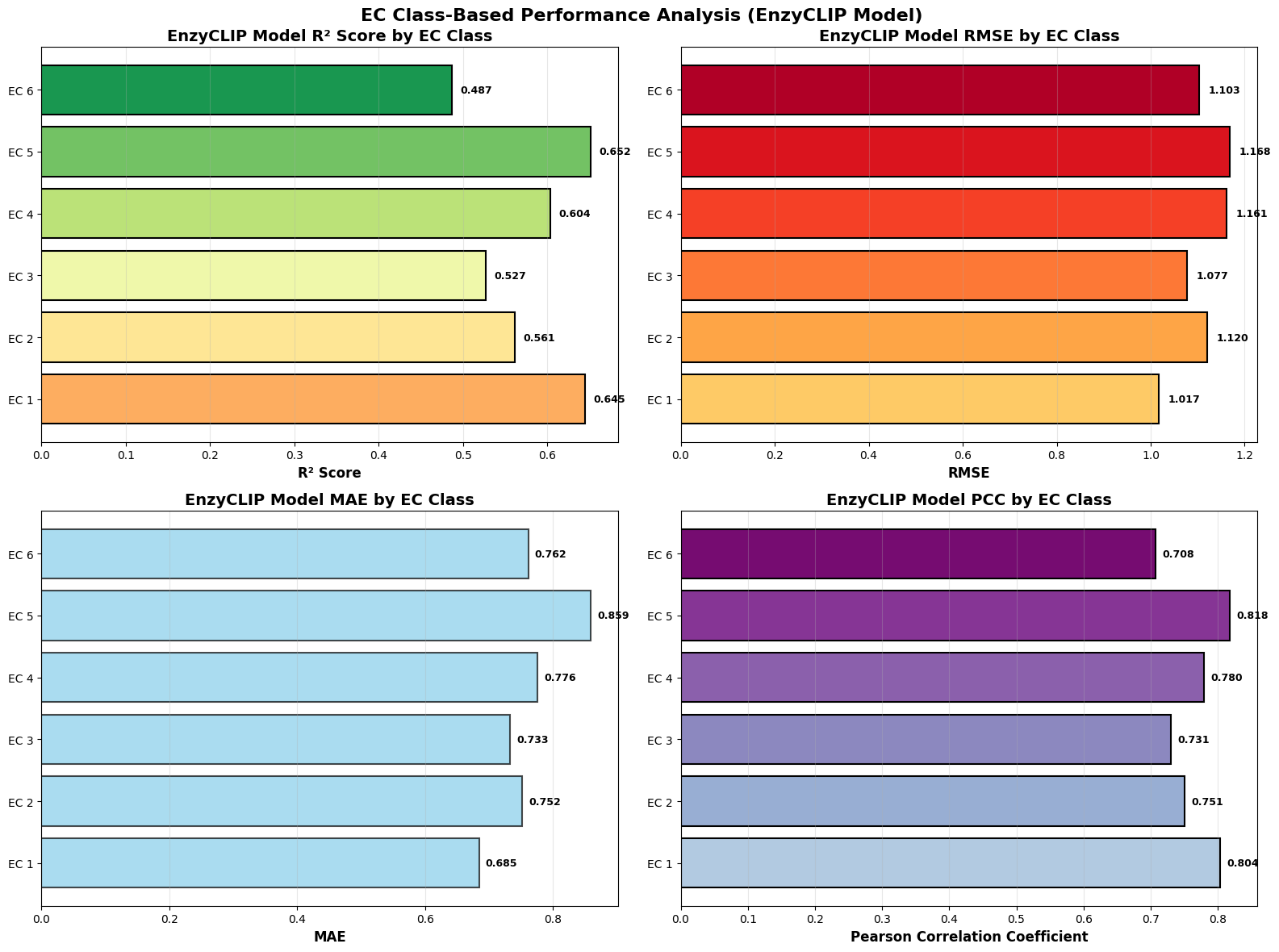
- EnzyCLIP achieves competitive baseline performance with R² scores of 0.593 for Kcat and 0.607 for Km prediction on the CatPred-DB dataset containing 23,151 Kcat and 41,174 Km measurements.
- The integration of contrastive learning with cross-attention mechanisms enables the model to capture biochemical relationships and substrate preferences even for unseen enzyme-substrate pairs.
- XGBoost ensemble methods applied to learned embeddings further improved Km prediction performance to R² = 0.61 while maintaining robust Kcat prediction capabilities.
摘要: Accurate prediction of enzyme kinetic parameters is crucial for drug discovery, metabolic engineering, and synthetic biology applications. Current computational approaches face limitations in capturing complex enzyme–substrate interactions and often focus on single parameters while neglecting the joint prediction of catalytic turnover numbers (Kcat) and Michaelis–Menten constants (Km). We present EnzyCLIP, a novel dual-encoder framework that leverages contrastive learning and cross-attention mechanisms to predict enzyme kinetic parameters from protein sequences and substrate molecular structures. Our approach integrates ESM-2 protein language model embeddings with ChemBERTa chemical representations through a CLIP-inspired architecture enhanced with bidirectional cross-attention for dynamic enzyme–substrate interaction modeling. EnzyCLIP combines InfoNCE contrastive loss with Huber regression loss to learn aligned multimodal representations while predicting log10-transformed kinetic parameters. EnzyCLIP is trained on the CatPred-DB database containing 23,151 Kcat and 41,174 Km experimentally validated measurements, and achieved competitive baseline performance with R2 scores of 0.593 for Kcat and 0.607 for Km prediction. XGBoost ensemble methods on learned embeddings further improved Km prediction (R2 = 0.61) while maintaining robust Kcat performance.