Paper List
-
A Unified Variational Principle for Branching Transport Networks: Wave Impedance, Viscous Flow, and Tissue Metabolism
This paper solves the core problem of predicting the empirically observed branching exponent (α≈2.7) in mammalian arterial trees, which neither Murray...
-
Household Bubbling Strategies for Epidemic Control and Social Connectivity
This paper addresses the core challenge of designing household merging (social bubble) strategies that effectively control epidemic risk while maximiz...
-
Empowering Chemical Structures with Biological Insights for Scalable Phenotypic Virtual Screening
This paper addresses the core challenge of bridging the gap between scalable chemical structure screening and biologically informative but resource-in...
-
A mechanical bifurcation constrains the evolution of cell sheet folding in the family Volvocaceae
This paper addresses the core problem of why there is an evolutionary gap in species with intermediate cell numbers (e.g., 256 cells) in Volvocaceae, ...
-
Bayesian Inference in Epidemic Modelling: A Beginner’s Guide Illustrated with the SIR Model
This guide addresses the core challenge of estimating uncertain epidemiological parameters (like transmission and recovery rates) from noisy, real-wor...
-
Geometric framework for biological evolution
This paper addresses the fundamental challenge of developing a coordinate-independent, geometric description of evolutionary dynamics that bridges gen...
-
A multiscale discrete-to-continuum framework for structured population models
This paper addresses the core challenge of systematically deriving uniformly valid continuum approximations from discrete structured population models...
-
Whole slide and microscopy image analysis with QuPath and OMERO
使QuPath能够直接分析存储在OMERO服务器中的图像而无需下载整个数据集,克服了大规模研究的本地存储限制。
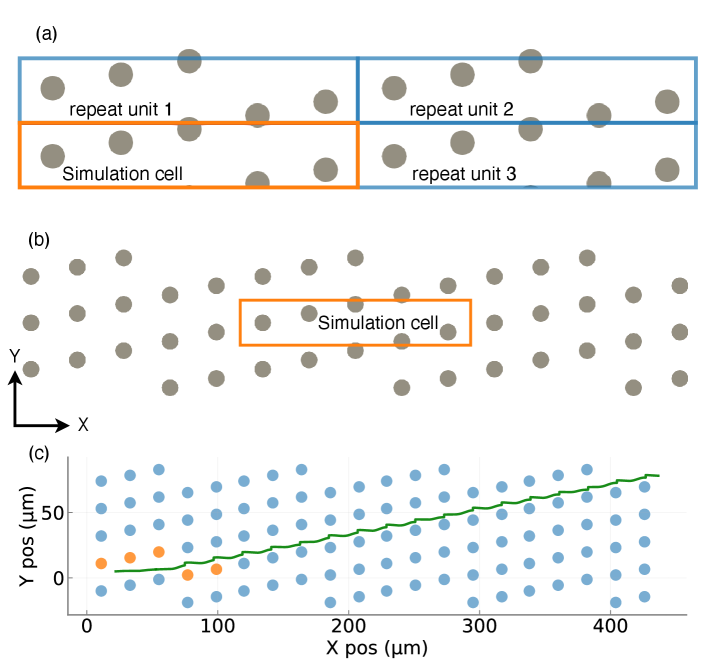
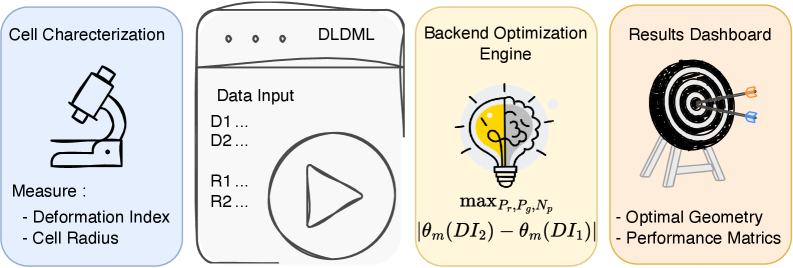
Physics-Guided Surrogate Modeling for Machine Learning–Driven DLD Design Optimization
Department of Mechanical Engineering, Lehigh University | Computational Engineering Department, Lawrence Livermore National Laboratory | Department of Industrial and Production Engineering, Bangladesh University of Engineering and Technology | Precision Medicine Translational Research Center, West China Hospital, Sichuan University
30秒速读
IN SHORT: This paper addresses the core bottleneck of translating microfluidic DLD devices from research prototypes to clinical applications by replacing weeks-long empirical design cycles with a physics-guided machine learning framework that delivers fabrication-ready specifications in under 60 seconds.
核心创新
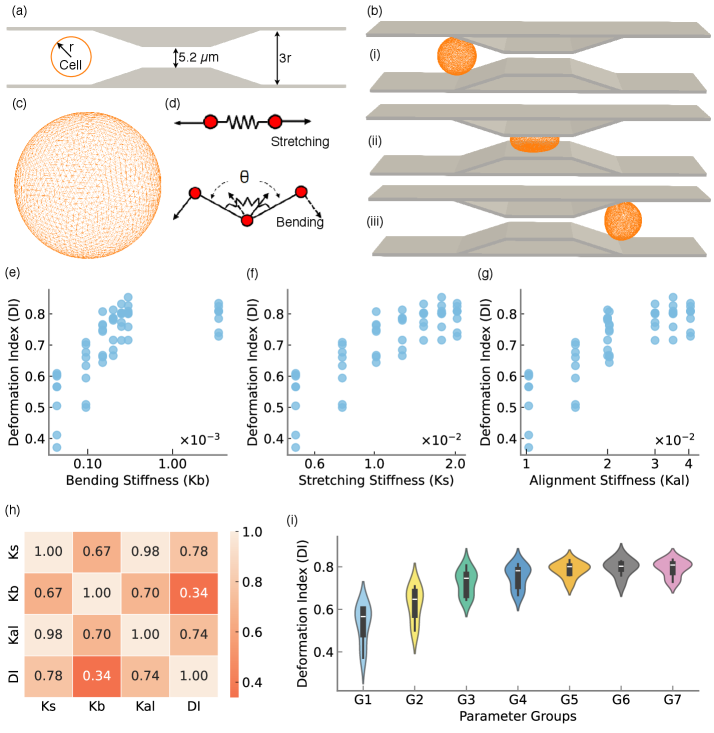
- Methodology First complete inverse design framework for DLD that transforms measured cellular deformability into optimized device geometry through physics-guided machine learning.
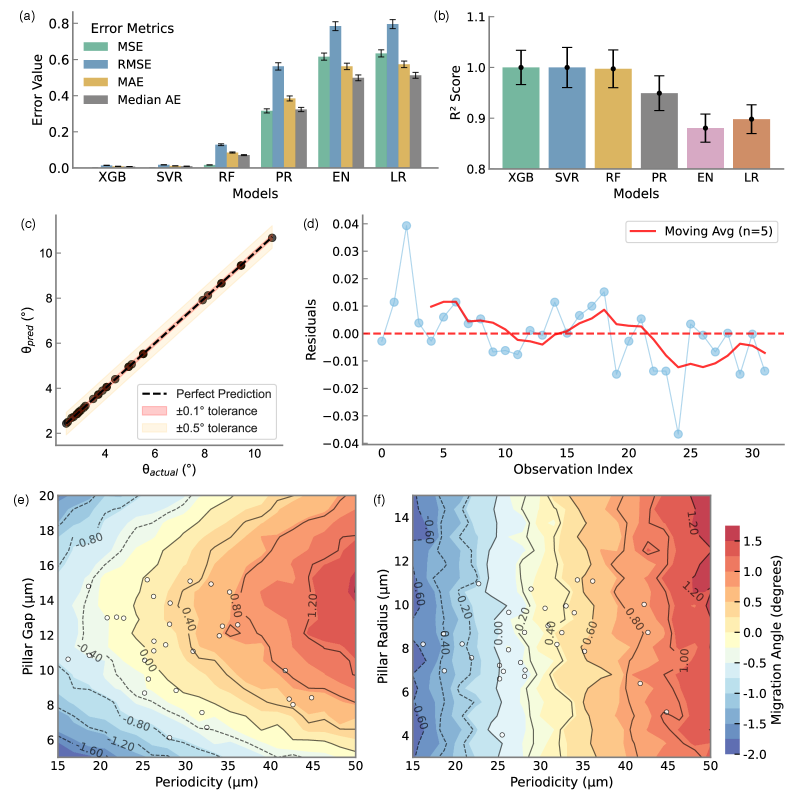
- Methodology Integration of high-fidelity Lattice-Boltzmann/Immersed-Boundary simulations with XGBoost surrogate models achieving sub-degree predictive accuracy (R²=0.9999, MSE=2×10⁻⁴).
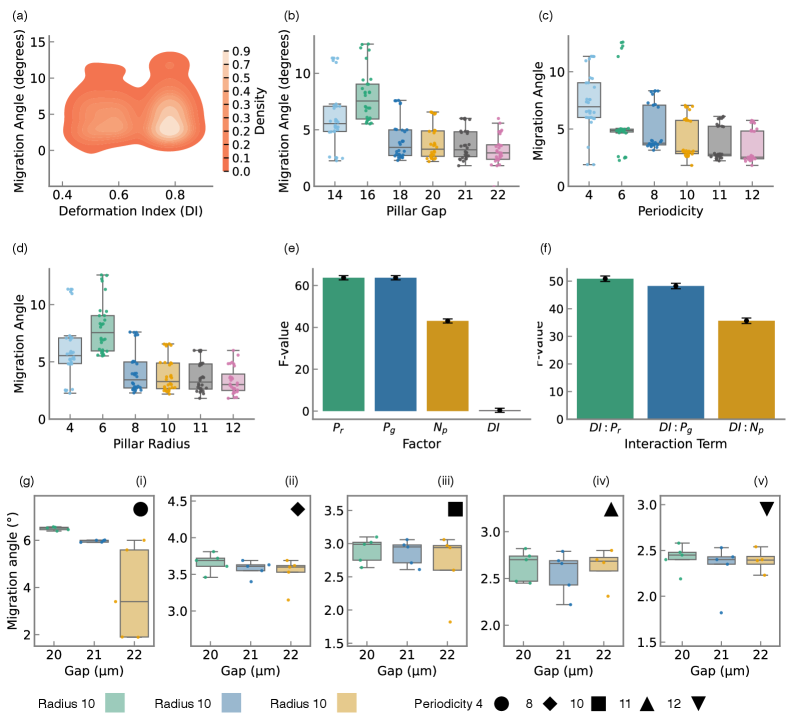
- Methodology Statistical quantification of deformability-geometry interactions via Type II ANOVA revealing significant interaction effects (F=48.23, p<10⁻³⁴) despite geometric dominance of main effects.
主要结论
- Geometric parameters dominate migration angle variance (F=63.72, p<10⁻³⁷), but cellular deformability exerts statistically significant effects through interactions with device geometry (F=48.23, p<10⁻³⁴).
- The XGBoost surrogate model achieves exceptional predictive accuracy (R²=0.9999, MSE=2×10⁻⁴), enabling sub-degree migration angle prediction across the design space.
- Bayesian optimization via tree-structured Parzen estimation identifies optimal DLD architectures in under 60 seconds, reducing design iteration from weeks of experimental prototyping to minutes of automated computation.
摘要: Microfluidic separation technologies have transformed label-free cell sorting by exploiting intrinsic biophysical properties, yet the translation of these platforms from laboratory prototypes to clinical applications remains constrained by the empirical, trial-and-error nature of device design. Deterministic Lateral Displacement (DLD) represents a paradigmatic example: while demonstrating robust discrimination of cells by size, shape, and deformability across diverse applications including circulating tumor cell isolation and malaria diagnostics, DLD performance exhibits extreme sensitivity to the coupled interplay between cellular mechanical phenotype and micron-scale geometric parameters, necessitating iterative fabrication-testing cycles that span weeks to months. We present the first complete inverse design framework that transforms measured cellular deformability into fabrication-ready DLD specifications through physics-guided machine learning. Our approach integrates high-fidelity lattice-Boltzmann and immersed-boundary simulations with gradient-boosted surrogate models to systematically map cellular mechanical properties to migration behavior across manufacturing-feasible geometric configurations (pillar radius, gap, periodicity). Type II ANOVA quantifies the relative influence of these parameters, revealing that while geometric factors dominate migration angle variance (F=63.72, p<10−37), cellular deformability exerts statistically significant effects through interactions with device geometry (F=48.23, p<10−34). The resulting XGBoost surrogate achieves sub-degree predictive accuracy (R2=0.9999, MSE =2×10−4), enabling Bayesian optimization via tree-structured Parzen estimation to identify optimal array architectures in under 60 seconds—reducing design iteration from weeks of experimental prototyping to minutes of automated computation. By deploying this validated pipeline as an accessible web application that accepts experimentally measured deformation indices and returns optimized device specifications with tolerance analysis, we democratize DLD design for researchers without specialized computational expertise, thereby accelerating the translation of microfluidic technologies from research-grade prototypes to application-specific, clinically deployable devices.