Paper List
-
Evolutionarily Stable Stackelberg Equilibrium
通过要求追随者策略对突变入侵具有鲁棒性,弥合了斯塔克尔伯格领导力模型与演化稳定性之间的鸿沟。
-
Recovering Sparse Neural Connectivity from Partial Measurements: A Covariance-Based Approach with Granger-Causality Refinement
通过跨多个实验会话累积协方差统计,实现从部分记录到完整神经连接性的重建。
-
Atomic Trajectory Modeling with State Space Models for Biomolecular Dynamics
ATMOS通过提供一个基于SSM的高效框架,用于生物分子的原子级轨迹生成,弥合了计算昂贵的MD模拟与时间受限的深度生成模型之间的差距。
-
Slow evolution towards generalism in a model of variable dietary range
通过证明是种群统计噪声(而非确定性动力学)驱动了模式形成和泛化食性的演化,解决了间接竞争下物种形成的悖论。
-
Grounded Multimodal Retrieval-Augmented Drafting of Radiology Impressions Using Case-Based Similarity Search
通过将印象草稿基于检索到的历史病例,并采用明确引用和基于置信度的拒绝机制,解决放射学报告生成中的幻觉问题。
-
Unified Policy–Value Decomposition for Rapid Adaptation
通过双线性分解在策略和价值函数之间共享低维目标嵌入,实现对新颖任务的零样本适应。
-
Mathematical Modeling of Cancer–Bacterial Therapy: Analysis and Numerical Simulation via Physics-Informed Neural Networks
提供了一个严格的、无网格的PINN框架,用于模拟和分析细菌癌症疗法中复杂的、空间异质的相互作用。
-
Sample-Efficient Adaptation of Drug-Response Models to Patient Tumors under Strong Biological Domain Shift
通过从无标记分子谱中学习可迁移表征,利用最少的临床数据实现患者药物反应的有效预测。
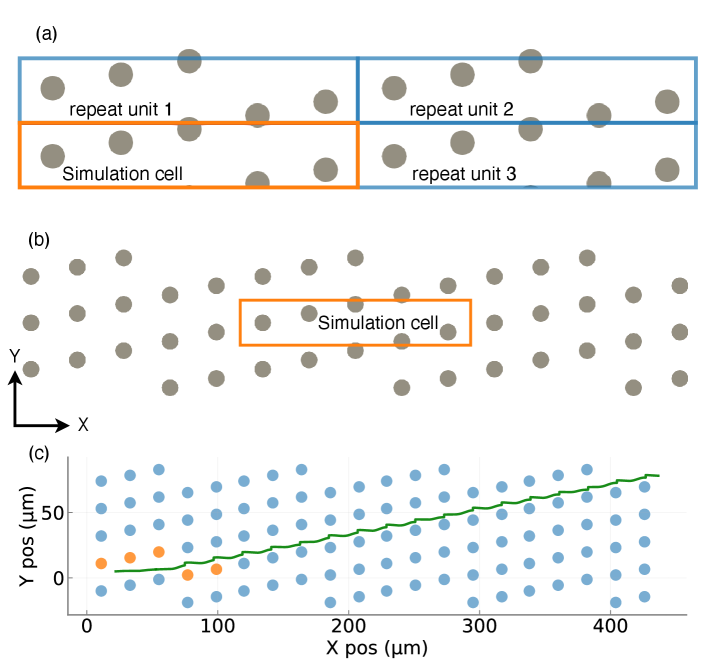
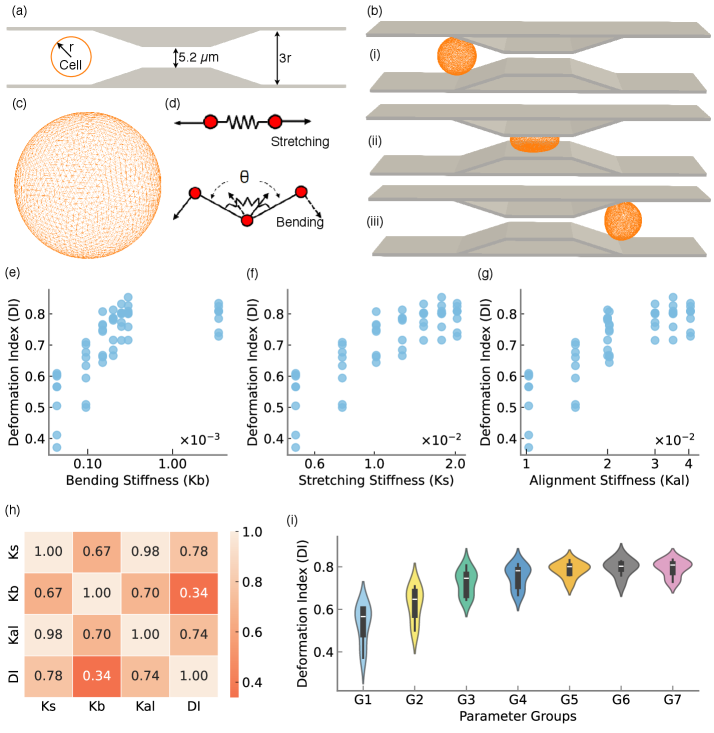
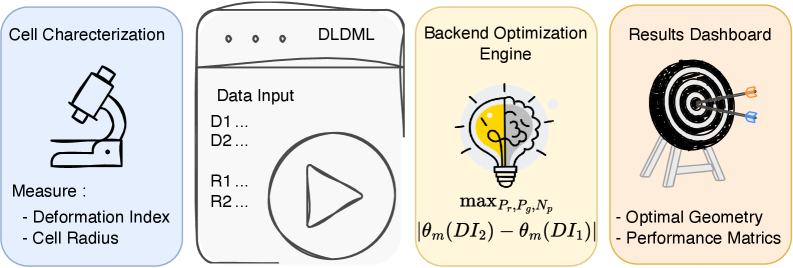
Physics-Guided Surrogate Modeling for Machine Learning–Driven DLD Design Optimization
Department of Mechanical Engineering, Lehigh University | Computational Engineering Department, Lawrence Livermore National Laboratory | Department of Industrial and Production Engineering, Bangladesh University of Engineering and Technology | Precision Medicine Translational Research Center, West China Hospital, Sichuan University
30秒速读
IN SHORT: This paper addresses the core bottleneck of translating microfluidic DLD devices from research prototypes to clinical applications by replacing weeks-long empirical design cycles with a physics-guided machine learning framework that delivers fabrication-ready specifications in under 60 seconds.
核心创新
- Methodology First complete inverse design framework for DLD that transforms measured cellular deformability into optimized device geometry through physics-guided machine learning.
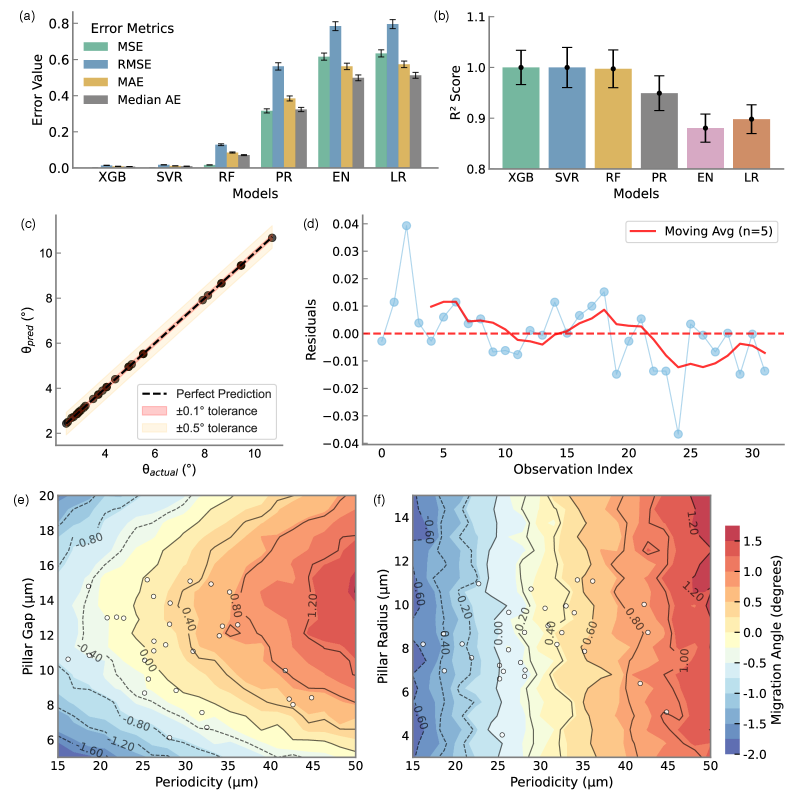
- Methodology Integration of high-fidelity Lattice-Boltzmann/Immersed-Boundary simulations with XGBoost surrogate models achieving sub-degree predictive accuracy (R²=0.9999, MSE=2×10⁻⁴).
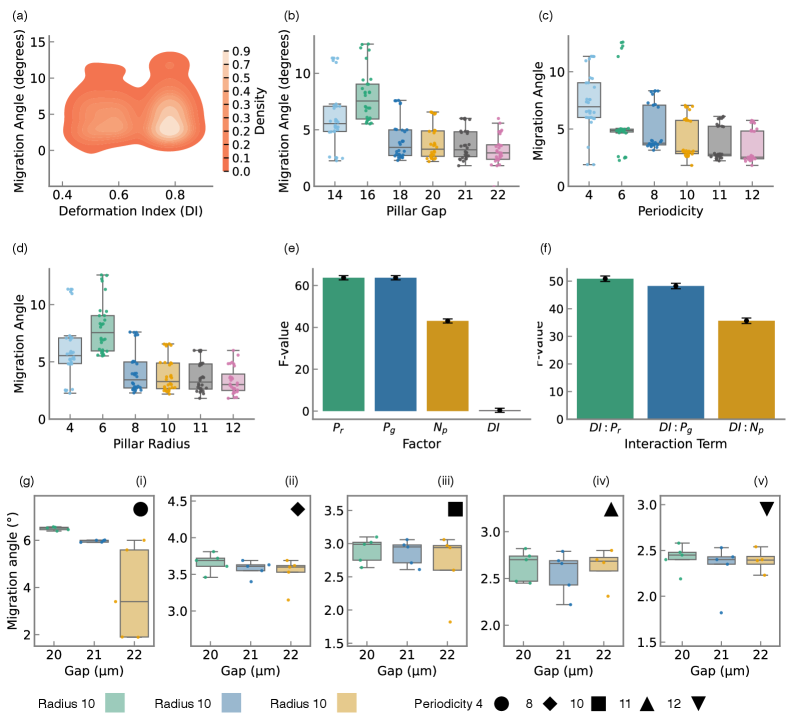
- Methodology Statistical quantification of deformability-geometry interactions via Type II ANOVA revealing significant interaction effects (F=48.23, p<10⁻³⁴) despite geometric dominance of main effects.
主要结论
- Geometric parameters dominate migration angle variance (F=63.72, p<10⁻³⁷), but cellular deformability exerts statistically significant effects through interactions with device geometry (F=48.23, p<10⁻³⁴).
- The XGBoost surrogate model achieves exceptional predictive accuracy (R²=0.9999, MSE=2×10⁻⁴), enabling sub-degree migration angle prediction across the design space.
- Bayesian optimization via tree-structured Parzen estimation identifies optimal DLD architectures in under 60 seconds, reducing design iteration from weeks of experimental prototyping to minutes of automated computation.
摘要: Microfluidic separation technologies have transformed label-free cell sorting by exploiting intrinsic biophysical properties, yet the translation of these platforms from laboratory prototypes to clinical applications remains constrained by the empirical, trial-and-error nature of device design. Deterministic Lateral Displacement (DLD) represents a paradigmatic example: while demonstrating robust discrimination of cells by size, shape, and deformability across diverse applications including circulating tumor cell isolation and malaria diagnostics, DLD performance exhibits extreme sensitivity to the coupled interplay between cellular mechanical phenotype and micron-scale geometric parameters, necessitating iterative fabrication-testing cycles that span weeks to months. We present the first complete inverse design framework that transforms measured cellular deformability into fabrication-ready DLD specifications through physics-guided machine learning. Our approach integrates high-fidelity lattice-Boltzmann and immersed-boundary simulations with gradient-boosted surrogate models to systematically map cellular mechanical properties to migration behavior across manufacturing-feasible geometric configurations (pillar radius, gap, periodicity). Type II ANOVA quantifies the relative influence of these parameters, revealing that while geometric factors dominate migration angle variance (F=63.72, p<10−37), cellular deformability exerts statistically significant effects through interactions with device geometry (F=48.23, p<10−34). The resulting XGBoost surrogate achieves sub-degree predictive accuracy (R2=0.9999, MSE =2×10−4), enabling Bayesian optimization via tree-structured Parzen estimation to identify optimal array architectures in under 60 seconds—reducing design iteration from weeks of experimental prototyping to minutes of automated computation. By deploying this validated pipeline as an accessible web application that accepts experimentally measured deformation indices and returns optimized device specifications with tolerance analysis, we democratize DLD design for researchers without specialized computational expertise, thereby accelerating the translation of microfluidic technologies from research-grade prototypes to application-specific, clinically deployable devices.