Paper List
-
Evolutionarily Stable Stackelberg Equilibrium
通过要求追随者策略对突变入侵具有鲁棒性,弥合了斯塔克尔伯格领导力模型与演化稳定性之间的鸿沟。
-
Recovering Sparse Neural Connectivity from Partial Measurements: A Covariance-Based Approach with Granger-Causality Refinement
通过跨多个实验会话累积协方差统计,实现从部分记录到完整神经连接性的重建。
-
Atomic Trajectory Modeling with State Space Models for Biomolecular Dynamics
ATMOS通过提供一个基于SSM的高效框架,用于生物分子的原子级轨迹生成,弥合了计算昂贵的MD模拟与时间受限的深度生成模型之间的差距。
-
Slow evolution towards generalism in a model of variable dietary range
通过证明是种群统计噪声(而非确定性动力学)驱动了模式形成和泛化食性的演化,解决了间接竞争下物种形成的悖论。
-
Grounded Multimodal Retrieval-Augmented Drafting of Radiology Impressions Using Case-Based Similarity Search
通过将印象草稿基于检索到的历史病例,并采用明确引用和基于置信度的拒绝机制,解决放射学报告生成中的幻觉问题。
-
Unified Policy–Value Decomposition for Rapid Adaptation
通过双线性分解在策略和价值函数之间共享低维目标嵌入,实现对新颖任务的零样本适应。
-
Mathematical Modeling of Cancer–Bacterial Therapy: Analysis and Numerical Simulation via Physics-Informed Neural Networks
提供了一个严格的、无网格的PINN框架,用于模拟和分析细菌癌症疗法中复杂的、空间异质的相互作用。
-
Sample-Efficient Adaptation of Drug-Response Models to Patient Tumors under Strong Biological Domain Shift
通过从无标记分子谱中学习可迁移表征,利用最少的临床数据实现患者药物反应的有效预测。
STAR-GO: Improving Protein Function Prediction by Learning to Hierarchically Integrate Ontology-Informed Semantic Embeddings
Department of Computer Engineering, Bogazici University, Istanbul, Turkiye
30秒速读
IN SHORT: This paper addresses the core challenge of generalizing protein function prediction to unseen or newly introduced Gene Ontology (GO) terms by overcoming the limitations of existing models that either prioritize graph structure at the expense of semantic meaning or vice versa.
核心创新
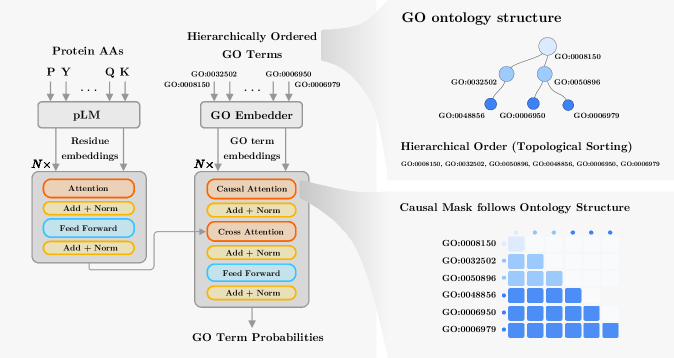
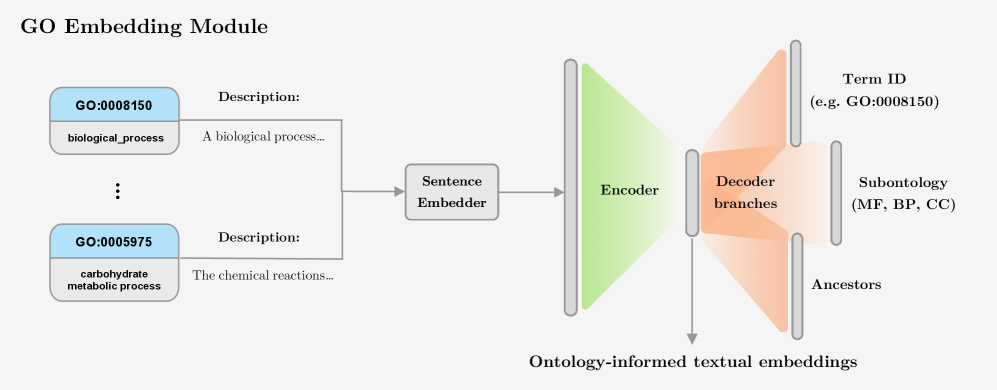
- Methodology Introduces a novel GO embedding module that integrates textual definitions (via SBERT-BioBERT) with ontology graph structure through a multi-task autoencoder, learning unified representations that preserve both semantic similarity and hierarchical dependencies.
- Methodology Proposes a hierarchical Transformer decoder that processes GO terms in topological order (ancestors to descendants) using causal self-attention, enabling information propagation across ontology levels and capturing functional dependencies.
- Biology Demonstrates superior zero-shot generalization to unseen GO terms, particularly for Molecular Function and Biological Process terms, by effectively leveraging semantic information from textual definitions, which transfers better to novel ontology concepts than purely structural embeddings.
主要结论
- STAR-GO achieves state-of-the-art or competitive performance across all three GO subontologies (BP, CC, MF), with the highest AUC scores (e.g., 0.989 for BP, 0.988 for CC, 0.995 for MF), indicating strong term-level discriminability.
- In zero-shot evaluation on 16 held-out GO terms, STAR-GO variants achieve the highest AUCs in 13 cases, significantly outperforming baselines like DeepGOZero and DeepGO-SE, demonstrating superior generalization to unseen functions.
- Ablation studies reveal that semantic embeddings (STAR_T) achieve the best zero-shot results for most MF and BP terms (e.g., AUC of 0.949 for GO:0001228), while structural embeddings (STAR_S) perform best for a few terms but poorly for MF, highlighting the critical role of semantic information for generalization.
摘要: Motivation: Accurate prediction of protein function is essential for elucidating molecular mechanisms and advancing biological and therapeutic discovery. Yet experimental annotation lags far behind the rapid growth of protein sequence data. Computational approaches address this gap by associating proteins with Gene Ontology (GO) terms, which encode functional knowledge through hierarchical relations and textual definitions. However, existing models often emphasize one modality over the other, limiting their ability to generalize, particularly to unseen or newly introduced GO terms that frequently arise as the ontology evolves, and making the previously trained models outdated. Results: We present STAR-GO, a Transformer-based framework that jointly models the semantic and structural characteristics of GO terms to enhance zero-shot protein function prediction. STAR-GO integrates textual definitions with ontology graph structure to learn unified GO representations, which are processed in hierarchical order to propagate information from general to specific terms. These representations are then aligned with protein sequence embeddings to capture sequence–function relationships. STAR-GO achieves state-of-the-art performance and superior zero-shot generalization, demonstrating the utility of integrating semantics and structure for robust and adaptable protein function prediction. Availability: Code and pre-trained models are available at https://github.com/boun-tabi-lifelu/stargo.