Paper List
-
A Unified Variational Principle for Branching Transport Networks: Wave Impedance, Viscous Flow, and Tissue Metabolism
This paper solves the core problem of predicting the empirically observed branching exponent (α≈2.7) in mammalian arterial trees, which neither Murray...
-
Household Bubbling Strategies for Epidemic Control and Social Connectivity
This paper addresses the core challenge of designing household merging (social bubble) strategies that effectively control epidemic risk while maximiz...
-
Empowering Chemical Structures with Biological Insights for Scalable Phenotypic Virtual Screening
This paper addresses the core challenge of bridging the gap between scalable chemical structure screening and biologically informative but resource-in...
-
A mechanical bifurcation constrains the evolution of cell sheet folding in the family Volvocaceae
This paper addresses the core problem of why there is an evolutionary gap in species with intermediate cell numbers (e.g., 256 cells) in Volvocaceae, ...
-
Bayesian Inference in Epidemic Modelling: A Beginner’s Guide Illustrated with the SIR Model
This guide addresses the core challenge of estimating uncertain epidemiological parameters (like transmission and recovery rates) from noisy, real-wor...
-
Geometric framework for biological evolution
This paper addresses the fundamental challenge of developing a coordinate-independent, geometric description of evolutionary dynamics that bridges gen...
-
A multiscale discrete-to-continuum framework for structured population models
This paper addresses the core challenge of systematically deriving uniformly valid continuum approximations from discrete structured population models...
-
Whole slide and microscopy image analysis with QuPath and OMERO
使QuPath能够直接分析存储在OMERO服务器中的图像而无需下载整个数据集,克服了大规模研究的本地存储限制。
PanFoMa: A Lightweight Foundation Model and Benchmark for Pan-Cancer
Extracted from affiliations in the content snippet (specific institutions not fully listed in provided text)
30秒速读
IN SHORT: This paper addresses the dual challenge of achieving computational efficiency without sacrificing accuracy in whole-transcriptome single-cell representation learning for pan-cancer analysis, moving beyond the limitations of pure Transformer or Mamba architectures.
核心创新
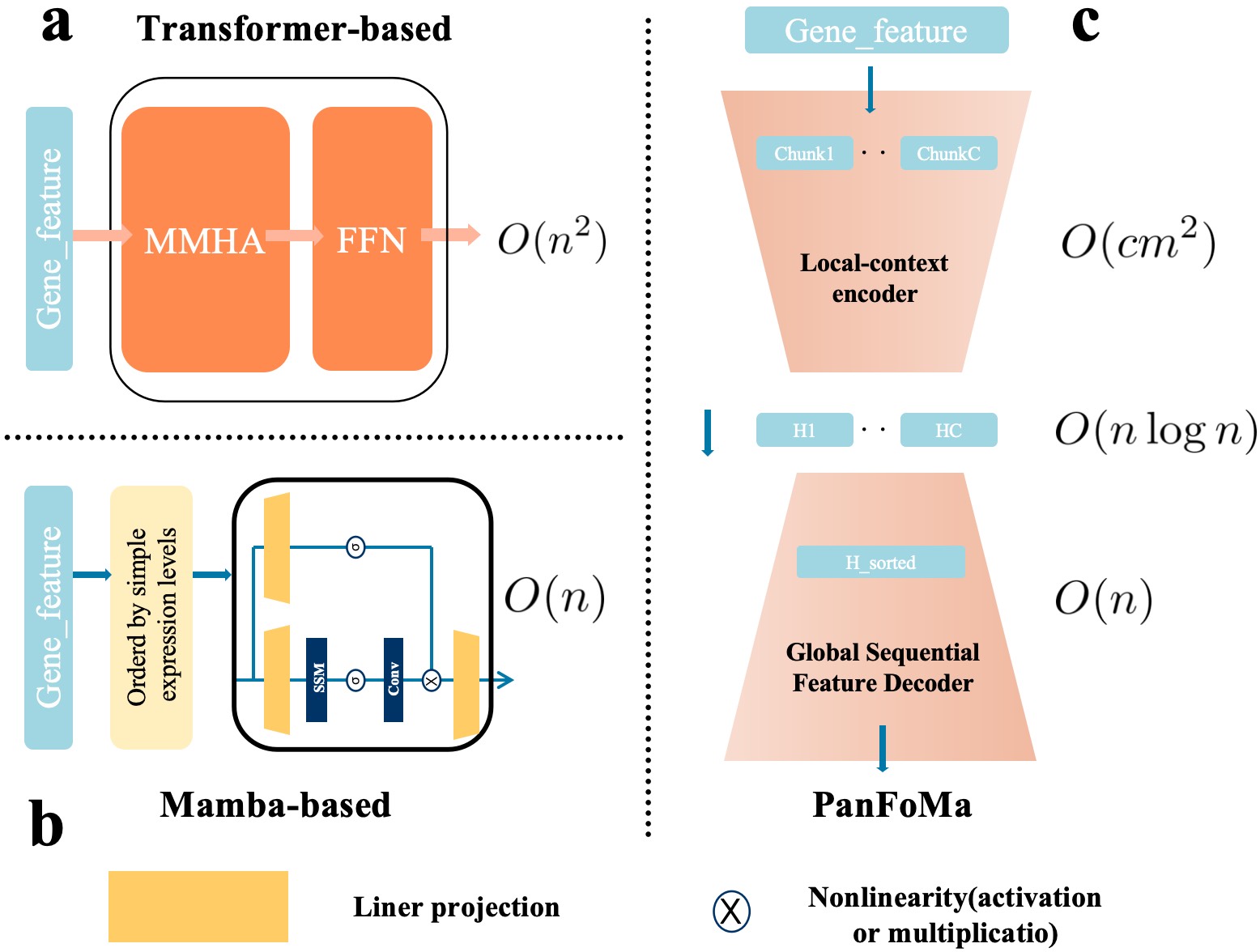
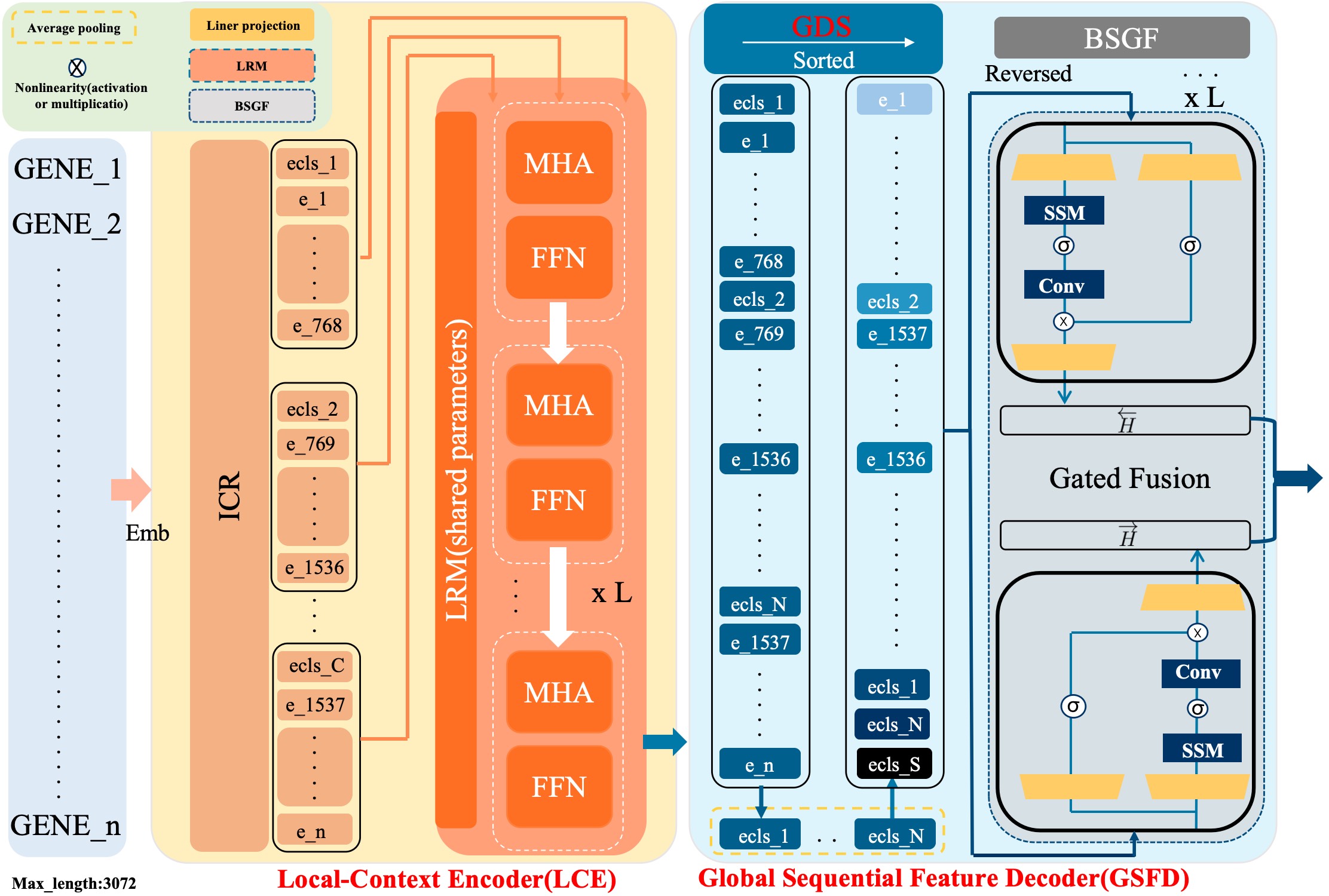
- Methodology Proposes a novel hybrid architecture (PanFoMa) that decouples local gene interaction modeling (via a lightweight, chunked Transformer encoder) from global context integration (via a bidirectional Mamba decoder), achieving O(C·M² + N log N) complexity.
- Methodology Introduces a Global-informed Dynamic Sorting (GDS) mechanism that adaptively orders genes for the Mamba decoder based on a learned global cell state vector, moving beyond static, heuristic gene ordering (e.g., by mean expression).
- Biology Constructs and releases PanFoMaBench, a large-scale, rigorously curated pan-cancer single-cell benchmark comprising over 3.5 million high-quality cells across 33 cancer subtypes from 23 tissues, addressing the lack of comprehensive evaluation resources.
主要结论
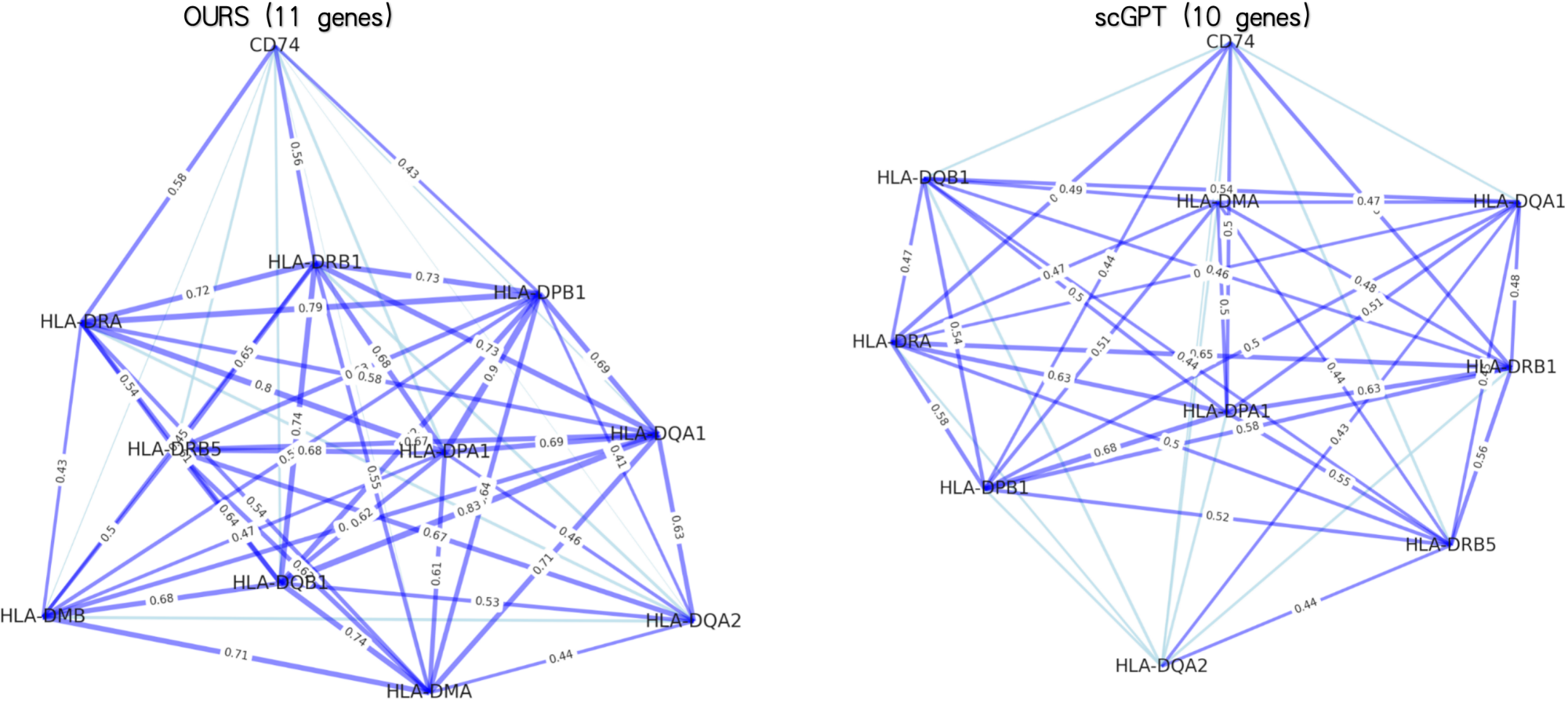
- PanFoMa achieves state-of-the-art pan-cancer classification accuracy of 94.74% (ACC) and 92.5% (Macro-F1) on PanFoMaBench, outperforming GeneFormer by +3.5% ACC and +4.0% F1.
- The model demonstrates superior generalizability across foundational tasks, showing improvements of +7.4% in cell type annotation, +4.0% in batch integration, and +3.1% in multi-omics integration over baselines.
- The hybrid local-global design and dynamic sorting are validated as effective, enabling efficient processing of full transcriptome-scale data (~3000 genes) while capturing both fine-grained local interactions and broad global regulatory patterns.
摘要: Single-cell RNA sequencing (scRNA-seq) is essential for decoding tumor heterogeneity. However, pan-cancer research still faces two key challenges: learning discriminative and efficient single-cell representations, and establishing a comprehensive evaluation benchmark. In this paper, we introduce PanFoMa, a lightweight hybrid neural network that combines the strengths of Transformers and state-space models to achieve a balance between performance and efficiency. PanFoMa consists of a front-end local-context encoder with shared self-attention layers to capture complex, order-independent gene interactions; and a back-end global sequential feature decoder that efficiently integrates global context using a linear-time state-space model. This modular design preserves the expressive power of Transformers while leveraging the scalability of Mamba to enable transcriptome modeling, effectively capturing both local and global regulatory signals. To enable robust evaluation, we also construct a large-scale pan-cancer single-cell benchmark, PanFoMaBench, containing over 3.5 million high-quality cells across 33 cancer subtypes, curated through a rigorous preprocessing pipeline. Experimental results show that PanFoMa outperforms state-of-the-art models on our pan-cancer benchmark (+4.0%) and across multiple public tasks, including cell type annotation (+7.4%), batch integration (+4.0%) and multi-omics integration (+3.1%). The code is available at https://github.com/Xiaoshui-Huang/PanFoMa.