Paper List
-
Developing the PsyCogMetrics™ AI Lab to Evaluate Large Language Models and Advance Cognitive Science
This paper addresses the critical gap between sophisticated LLM evaluation needs and the lack of accessible, scientifically rigorous platforms that in...
-
Equivalence of approximation by networks of single- and multi-spike neurons
This paper resolves the fundamental question of whether single-spike spiking neural networks (SNNs) are inherently less expressive than multi-spike SN...
-
The neuroscience of transformers
提出了Transformer架构与皮层柱微环路之间的新颖计算映射,连接了现代AI与神经科学。
-
Framing local structural identifiability and observability in terms of parameter-state symmetries
This paper addresses the core challenge of systematically determining which parameters and states in a mechanistic ODE model can be uniquely inferred ...
-
Leveraging Phytolith Research using Artificial Intelligence
This paper addresses the critical bottleneck in phytolith research by automating the labor-intensive manual microscopy process through a multimodal AI...
-
Neural network-based encoding in free-viewing fMRI with gaze-aware models
This paper addresses the core challenge of building computationally efficient and ecologically valid brain encoding models for naturalistic vision by ...
-
Scalable DNA Ternary Full Adder Enabled by a Competitive Blocking Circuit
This paper addresses the core bottleneck of carry information attenuation and limited computational scale in DNA binary adders by introducing a scalab...
-
ELISA: An Interpretable Hybrid Generative AI Agent for Expression-Grounded Discovery in Single-Cell Genomics
This paper addresses the critical bottleneck of translating high-dimensional single-cell transcriptomic data into interpretable biological hypotheses ...
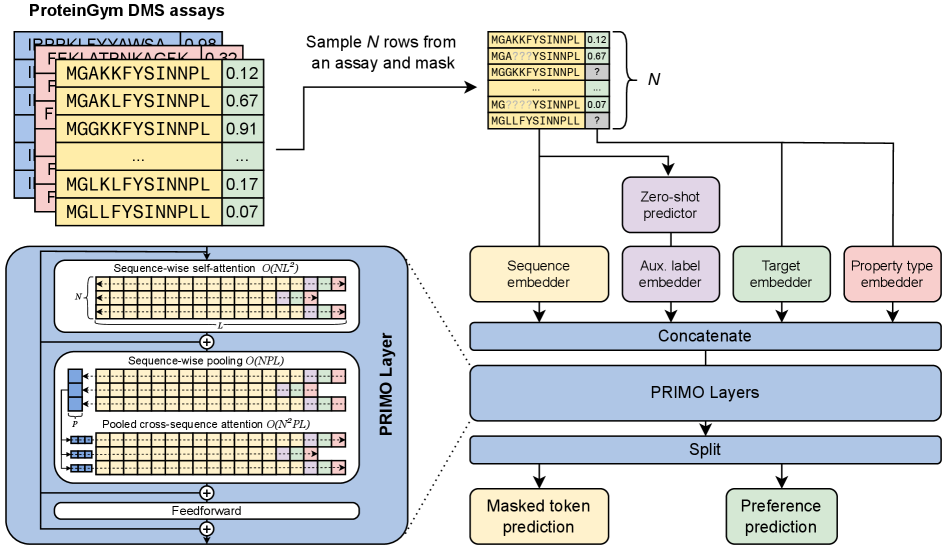
Few-shot Protein Fitness Prediction via In-context Learning and Test-time Training
Department of Systems Biology, Harvard Medical School | Department of Biology, University of Copenhagen | Machine Intelligence, Novo Nordisk A/S | Microsoft Research, Cambridge, MA, USA | Dept. of Applied Mathematics and Computer Science, Technical University of Denmark
30秒速读
IN SHORT: This paper addresses the core challenge of accurately predicting protein fitness with only a handful of experimental observations, where data collection is prohibitively expensive and label availability is severely limited.
核心创新
- Methodology Introduces PRIMO, a novel transformer-based framework that uniquely combines in-context learning with test-time training for few-shot protein fitness prediction.
- Methodology Proposes a hybrid masked token reconstruction objective with a preference-based loss function, enabling effective learning from sparse experimental labels across diverse assays.
- Methodology Develops a lightweight pooling attention mechanism that handles both substitution and indel mutations while maintaining computational efficiency, overcoming limitations of previous methods.
主要结论
- PRIMO with test-time training (TTT) achieves state-of-the-art few-shot performance, improving from a zero-shot Spearman correlation of 0.51 to 0.67 with 128 shots, outperforming Gaussian Process (0.56) and Ridge Regression (0.63) baselines.
- The framework demonstrates broad applicability across protein properties including stability (0.77 correlation with TTT), enzymatic activity (0.61), fluorescence (0.30), and binding (0.69), handling both substitution and indel mutations.
- PRIMO's performance highlights the critical importance of proper data splitting to avoid inflated results, as demonstrated by the 0.4 correlation inflation on RL40A_YEAST when using Metalic's overlapping train-test split.
摘要: Accurately predicting protein fitness with minimal experimental data is a persistent challenge in protein engineering. We introduce PRIMO (PRotein In-context Mutation Oracle), a transformer-based framework that leverages in-context learning and test-time training to adapt rapidly to new proteins and assays without large task-specific datasets. By encoding sequence information, auxiliary zero-shot predictions, and sparse experimental labels from many assays as a unified token set in a pre-training masked-language modeling paradigm, PRIMO learns to prioritize promising variants through a preference-based loss function. Across diverse protein families and properties—including both substitution and indel mutations—PRIMO outperforms zero-shot and fully supervised baselines. This work underscores the power of combining large-scale pre-training with efficient test-time adaptation to tackle challenging protein design tasks where data collection is expensive and label availability is limited.